Skyhawk Therapeutics Appoints Aaron Deves as Chief Commercial Officer to Lead Commercialization of SKY-0515 for Huntington's Disease
Industry veteran brings more than 30 years of experience launching therapies for neurological diseases, including Huntington's disease, and will prepare Skyhawk for the launch of SKY-0515 as early as 2027.
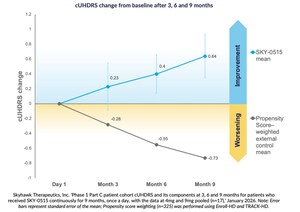
BOSTON, March 10, 2026 /PRNewswire/ -- Skyhawk Therapeutics, Inc., a clinical-stage biotechnology company developing novel small molecule therapies to modulate critical RNA targets for a series of challenging neurological diseases, announces the appointment of Aaron Deves as Chief Commercial Officer. Mr. Deves brings more than 30 years of experience commercializing therapies for neurological disorders, including for chorea associated with Huntington's disease (HD) with AUSTEDO®. SKY-0515 is Skyhawk's lead program and is being developed as a potential disease-modifying therapy for Huntington's disease.
"Skyhawk may receive accelerated approval for SKY-0515 in Australia within the next twelve months, and in other major markets during 2027," said Bill Haney, CEO of Skyhawk Therapeutics. "Aaron's 30 years of experience successfully building commercial teams and launching innovative drugs for challenging neurological conditions helps prepare Skyhawk to bring a much-needed disease-modifying treatment to patients with Huntington's disease as quickly as possible - pending additional clinical results and regulatory approvals."
"I am incredibly excited to join Skyhawk," said Aaron Deves, Chief Commercial Officer of Skyhawk Therapeutics. "Skyhawk's Huntington's disease program can be the cornerstone of a powerful commercial neuro business. And the company's rich pre-clinical pipeline of RNA targeting drug programs addresses the most impressive set of challenging neurological conditions I've seen in my career – and does so with small molecules that are often the most patient friendly modality. I'm thrilled to join the company to prepare for the launch of SKY-0515 and to help ensure the broadest number of patients can access this important therapy."
About Aaron Deves
Aaron Deves is an accomplished biopharmaceutical executive with 30 years of experience focusing on commercializing novel treatments for challenging neurological diseases.
Most recently Aaron served as Senior Vice President, Chief Operating Officer, and Head of Marketing for the U.S. Specialty Business at Teva Pharmaceuticals where he led a commercial team of approximately 800 employees across marketing, sales, market access, and medical affairs to sell multiple products, including AUSTEDO® for chorea associated with Huntington's disease.
Prior to Teva, Aaron held senior leadership roles at Otsuka Pharmaceuticals and Pfizer. At Otsuka he served as Vice President of U.S. and Global Marketing leading the commercial strategy for the neuroscience portfolio, including all commercial aspects of ABILIFY®, REXULTI®, and the development of the company's Alzheimer's portfolio.
Aaron began his career at Pfizer, where he spent 16 years in progressively senior roles across marketing, sales, operations, and global development, including contributing to the global development of Alzheimer's disease therapies. Aaron holds a B.S. in Business Administration and Marketing from Columbia College.
About Skyhawk Therapeutics
Skyhawk Therapeutics is a clinical-stage biotechnology company which uses its proprietary platform, SKYSTAR®, to discover and develop small molecule RNA modulating therapies for the world's most intractable diseases. For more information visit, www.skyhawktx.com.
Skyhawk Contact
Maura McCarthy
Head of Corporate Development
[email protected]





Artikel teilen