TruLeaf Medical, a fully-owned subsidiary of Allmed Solutions successfully Completed the Second Stage of valve implantation in two Additional Patients
In a follow up of up to nine months, TruLeaf Proprietary RoseDoc Docking System Demonstrates Exceptional Clinical Safety and Efficacy for transcatheter tricuspid valve replacement.
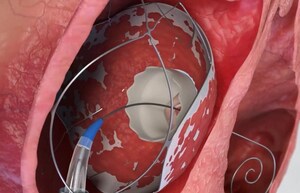
OR YEHUDA, Israel, May 4, 2026 /PRNewswire/ -- TruLeaf Medical, Ltd, a fully-owned subsidiary of Allmed Solutions (TASE: ALMD), a clinical-stage medical device company specializing in transcatheter structural heart innovations, is pleased to announce the successful completion of the full two-stage percutaneous heart valve implantation procedure in two additional high-risk patients suffering from severe, refractory heart failure due to leaky tricuspid valve (TR). The first stage of the docking device implantation was performed 3 months ago. In recent days, the second stage of fully percutaneous transcatheter heart valve implantation within the TruLeaf RoseDoc docking station, was successfully performed at a leading medical center in India under a compassionate-use framework. This milestone achievement, conducted in collaboration with a prominent international medical device partner, further validates the clinical utility of TruLeaf Medical RoseDoc TTVR system for high-risk, no-other-option patients suffering from severe refractory heart failure due to severe TR.
This latest milestone builds upon the company's landmark first-in-human procedures reported in September and December of 2025. In up to nine-months follow-up of the initial four cases, the RoseDoc platform demonstrates exceptional efficacy, safety and long-term stability, confirming the TruLeaf system's unique ability to provide a secure and permanent 'landing zone' for heart valves. By creating a stable, permanent anchor within the heart's anatomy, the system effectively addresses the primary challenges of transcatheter valve replacement for both the tricuspid and mitral valves, including valve migration, paravalvular leaks, heart-block requiring pacemaker implantation (in TTVR) and left ventricular outflow tract obstruction (in TMVR), ensuring optimal clinical and hemodynamic results for high-risk patients.
This method expands life-saving treatment options for patients who were previously considered untreatable. "The successful implantation in these additional patients, combined with the exceptional ongoing performance of our initial cases, marks a pivotal moment for Allmed Solutions," said Professor Oz Shapira, CEO of Allmed Solutions. "Our TruLeaf docking system is proving to be a true game-changer, providing the stability and precision required to ensure the long-term success of transcatheter valve replacements. We are excited to see our technology consistently delivering life-saving results in collaboration with world-class international partners."
Photo: https://mma.prnewswire.com/media/2971796/AllMeD_Solutions_Simulation.jpg
SOURCE AllMeD Solutions





Share this article