
Nationwide overnight delivery from Walmart improves access to novel at-home prescription treatment, with particular benefit for rural communities where transportation and distance are often barriers to timely treatment.
BURLINGTON, Mass., April 21, 2026 /PRNewswire/ -- SQ Innovation, developer of Lasix ONYU, today announced a collaboration with Walmart Specialty Pharmacy to expand nationwide access to this recently introduced subcutaneous furosemide treatment for patients with heart failure-related edema.
Edema in heart failure is the second most common reason for hospital admission among elderly patients1, placing significant personal and financial burden on patients, families, and the healthcare system. Through this collaboration, Walmart Specialty Pharmacy will provide nationwide overnight delivery of Lasix ONYU.
Access to Lasix ONYU, available by prescription, is of particular importance for rural America, where travel times to medical facilities are often long and many patients face transportation and mobility challenges. By having the option for treatment that is directly and quickly to patients' homes through Walmart Specialty Pharmacy's overnight delivery capabilities, barriers to parenteral diuretics are significantly reduced. Such treatments are now typically limited to hospitals and other medical facilities.
"Lasix ONYU is an enabler for cost-effective at-home treatment of fluid overload, but a product alone does not change a 60-year-old important clinical paradigm" said Pieter Muntendam MD founder, President and CEO of SQ Innovation. "It needs a broad collaboration between all parties and Walmart with its nationwide reach and pharmacy capabilities, contributes to expanding patient access helping to establish the home as an alternative and in the future possibly the preferred place for treatment of edema in heart failure for most patients."
SQ Innovation is working with payers to establish a reimbursement framework that minimizes barriers for this treatment, which aims to reduce the personal and financial burden of inpatient care for heart failure patients and their families. Patients interested in overnight delivery of Lasix ONYU, through Walmart Specialty Pharmacy are encouraged to contact the pharmacy to discuss available payment options.
About Lasix ONYU
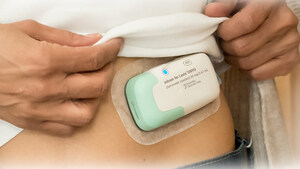
Lasix® ONYU is a drug-device combination that was approved by the U.S. Food and Drug Administration on October 7, 2025 for the treatment of edema in adult patients with chronic heart failure. The pharmaceutical component of Lasix ONYU is a novel, high-concentration formulation of the diuretic furosemide, at 30 mg/mL. It comes in a pre-filled glass cartridge containing 80 mg of furosemide in 2.67 mL. The Lasix ONYU Infusor consists of two main parts: the Reusable Unit and the Disposable Unit. The Reusable Unit is an electromechanical device that contains the battery, motor, and electronic components necessary for operation and safety functions. It can be used up to 48 times before it can be recycled. The Disposable Unit is a sterile, single-use plastic component that holds the drug cartridge. It includes a micropiston pump, fluid path, needle insertion and retraction mechanism, and a 29-gauge needle. After placement on the abdomen, the needle penetrates the skin when the device is activated. The Lasix ONYU Infusor slowly administers 80 mg furosemide over a period of five hours. This method results in significant diuresis similar to IV, but in a more controlled manner. This avoids the brief, intense diuretic effect that occurs with rapid IV infusion or injection. The advanced two component design offers benefits for patients, healthcare providers, payers, and the environment. For Important Safety Information, Prescribing Information and Instructions for Use, visit www.lasix-onyu.com.
INDICATION
Lasix® ONYU is a loop diuretic indicated for the treatment of edema in adult patients with chronic heart failure.
IMPORTANT SAFETY INFORMATION
Before using Lasix® ONYU, read the Instructions for Use and tell your healthcare provider about all your medical conditions, including if you are allergic to furosemide or any of the ingredients in Lasix ONYU, have trouble urinating, or if you are allergic to medical adhesives.
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines.
Warning: Only use the Lasix ONYU Prefilled Cartridge with the Lasix ONYU Infusor. Do not use insulin cartridges or other medicine cartridges in the Lasix ONYU Infusor. Doing so could cause severe injury.
What are the possible side effects of Lasix ONYU?
Dehydration: Lasix ONYU is a diuretic that can make you lose a lot of fluid and with it electrolytes. You may get a dry mouth, have increased thirst, get muscle pains or cramps, have reduced urine output or your urine may be more yellow than normal, you may get a headache, get dry skin, or have nausea or vomiting. Your healthcare provider may check your electrolytes while receiving Lasix ONYU.
Low Blood Pressure: Lasix ONYU may lower your blood pressure temporarily. You may feel lightheaded or dizzy. This usually happens when you stand. Getting up slowly may help.
High Blood Sugar: Lasix ONYU may increase blood sugar (glucose) levels if you have diabetes mellitus.
Loss of Hearing: Lasix ONYU can cause ringing in your ears. If so, tell your healthcare provider.
Risk of Sunburn: Your skin may be more sensitive to sunlight while taking Lasix ONYU.
Infusion Site Reactions: Lasix ONYU can cause infusion site pain, bruising and temporary swelling or redness at the site of the Infusor.
Incomplete Dosing: Make sure the Infusor does not get wet during use. Also limit your physical activities. Some movements or when it gets wet may stop the infusion and you may not get all the medication.
These are not all the possible side effects of Lasix ONYU. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
Please see the full Prescribing Information and Instructions for Use at www.lasix-onyu.com.
About SQ Innovation
SQ Innovation, Inc. is a privately held Swiss biopharmaceutical company with offices in Zug, Switzerland, Burlington, MA, USA, and Rotterdam, The Netherlands. The company was founded to develop and commercialize innovative, cost-effective therapies for subcutaneous delivery, enabling at-home treatment for conditions that are usually managed during hospitalizations. SQ Innovation has developed a novel drug-device combination for treating edema in adult patients with chronic heart failure — a condition typically requiring intravenous administration of diuretics in a hospital setting. Lasix ONYU was developed with consideration for patients, payors, healthcare providers, and environmental impact. Lasix ONYU received approval from the US Food and Drug Administration on October 7, 2025. For more information about Lasix ONYU, including important safety information and the full prescribing information, please visit www.lasix-onyu.com.
For Media Inquiries:
[email protected]
1 Definitive Healthcare 2024 Medicare Claims Data
SOURCE SQ Innovation







Share this article