
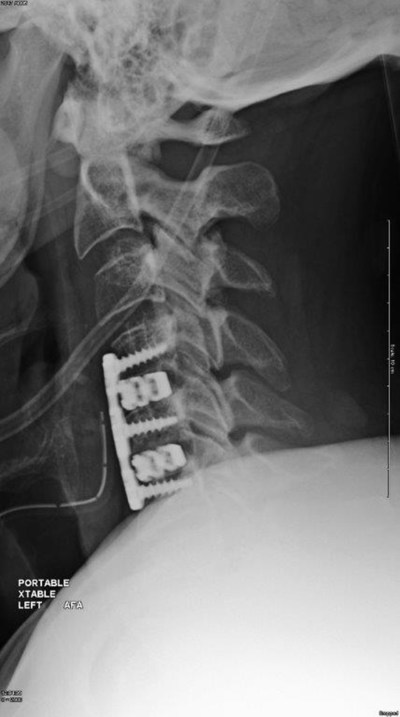
WAYNE, Pa., Feb. 12, 2018 /PRNewswire/ -- Camber Spine, a leading innovator in spine and medical technologies, today announced the first surgeries using the company's proprietary SPIRA™-C Open Matrix Cervical Interbody device, a unique, interbody fusion implant consisting of spiral support arches and Surface by Design™ technology. The first cases were performed successfully this past week with several surgeons across the country performing multilevel ACDF procedures.
Dr. Michael Dorsi, M.D. of Ventura Neurosurgery was one of the first surgeon design team members to perform a multilevel procedure using SPIRA™-C this past week. According to Dr. Dorsi: "I really appreciate the design of this implant. Upon insertion, I feel as though the cage has immediate stability because of the roughened surface which provides significant friction. The arched design has large openings which allows for significant graft packing and creates a snowshoe-like effect of distributed load sharing which should decrease the risk of subsidence. The open matrix architecture eliminates the need for the central lumen in traditional cervical cages, potentially speeding up the mechanical fusion process, optimizing clinical and fusion outcomes."
Dr. Michael Dorsi, M.D., is a board-certified, fellowship trained neurosurgeon who has been providing state-of-the-art care to the greater Ventura County area since 2012. Dr. Dorsi attended The Johns Hopkins University, graduated Phi Beta Kappa with Highest Honors and a degree in Neuroscience and subsequently completed his residency training in neurosurgery. He was awarded the Cloward Award from the American Association of Neurological Surgeons for Fellowship Training in Minimally and Invasive Spine Surgery at UCLA.
SPIRA™-C was designed to address surgeon frustrations associated with traditional cervical interbody devices such as the need for immediate stability. SPIRA™-C was developed for Camber Spine under a collaborative agreement with the Institute for Musculoskeletal Science & Education (IMSE), an independent, full-service product development and engineering services company. With an extensive faculty of dedicated physicians, IMSE leverages the collaborative balance between engineering and surgeon ingenuity to create next-level medical devices. The SPIRA™-C design team included several John Hopkins trained Neurosurgeons, all with extensive backgrounds treating complicated cervical pathologies.
"Surgeon design team members are a key component to our success. The insight they provide helps us create smarter solutions, maximizing technological advances and ultimately driving innovation in minimally invasive spine surgery. We are fortunate to have an esteemed surgeon such as Dr. Dorsi on our team helping us turn surgeon frustrations into innovations. There is a lot of excitement around the SPIRA™ technology platform and we expect products like SPIRA™-C and SPIRA™ALIF to fuel our continued sales growth."
- Seth Anderson, Founding Partner and Vice President, Surgeon Relations
SPIRA™-C is the second in the SPIRA™ family to employ a novel arched design as well as Surface by Design™ technology specifically designed to increase fusion rates and stabilization. The spiral support arches decrease subsidence by load sharing over the entire endplate, while also maximizing bone graft capacity. The Surface by Design™ technology is a deliberately designed roughened surface that facilitates bone growth through an optimized pore diameter, strut thickness and trabecular pattern.
About Camber Spine
Camber Spine Technologies, LLP, is a medical device company focused on the design, development and commercialization of innovative and proprietary musculoskeletal implant systems. The company is committed to delivering surgeon inspired new technologies to the spine market. Camber Spine, located in Wayne, Pennsylvania, markets a line of proprietary musculoskeletal products nationwide through its exclusive distributor, S1 Spine.
All of Camber Spine's products are proudly MADE IN THE USA.
SOURCE Camber Spine Technologies


Share this article