
- 53% 1-year Overall Survival and 32% 2-year Overall Survival are approximately double outcomes with current approaches
- Patients on study received a median of 2 lines of prior therapy with 57% receiving prior treatment with venetoclax and 67% had adverse cytogenetics with 52% of patients having a TP53 mutation
- Data accepted for oral presentation at the 64th American Society of Hematology (ASH) Annual Meeting & Symposium on Saturday, December 10th at 10:30 AM CT
NEW YORK, Nov. 3, 2022 /PRNewswire/ -- Actinium Pharmaceuticals, Inc. (NYSE AMERICAN: ATNM) (Actinium or the Company) a leader in the development of targeted radiotherapies, today highlighted positive results from the Phase 1 trial evaluating Actimab-A with salvage chemotherapy regimen CLAG-M in fit patients with relapsed or refractory acute myeloid leukemia (AML) that is being conducted at the Medical College of Wisconsin as investigator sponsored study. These data were detailed in an abstract accepted for oral presentation at the 64th Annual ASH Meeting & Symposium being held December 10-13, 2022 in New Orleans, Louisiana.
- Median Overall Survival (OS) of 12 months amongst all patients (n=21)
- 1-year OS of 53% and 2-year OS of 32%
- Overall Response Rate (ORR) of 67% across all dose cohorts
- 72% MRD negativity rate in patients achieving CRc
- ORR of 83% at the recommended Phase 2 dose of 0.75uCi/kg of Actimab-A with CLAG-M
- Actimab-A CLAG-M combo was active in patients with TP53 mutations with an ORR of 73% and an ORR of 55% in patients previously treated with venetoclax
Dr. Sameem Abedin, Assistant Professor at Froedtert & Medical College Wisconsin and Principal Investigator of the Study, commented, "The median overall survival of 12 months and 2-year overall survival of 32% is highly impressive in these relapsed or refractory patients, where a majority of treated patients have adverse cytogenetics including TP53 mutations and received prior venetoclax therapy. These are extremely difficult to treat patients with very limited treatment options and their expected median overall survival is approximately 2 to 3 months."
"These data strongly support the further clinical development of this novel targeted radiotherapy-based combination. We are excited that our hypothesis of adding Actimab-A to CLAG-M to eliminate residual leukemia cells, resulting in deeper remissions and survival with acceptable tolerability given targeted nature of Actimab-A is strongly supported by these findings," concluded Dr. Abedin.
- Patients had relapsed or refractory AML and deemed fit with adequate organ function
- Patients received a median of two lines of prior therapy
- 57% received prior venetoclax therapy
- 67% of patients had adverse cytogenetics, 52% had TP53 mutations
- Median age was 63 years
- Patients had median blast CD33 expression of 77% (>25% required for enrollment)
- 52% of patients had secondary AML or treatment related AML
Dr. Avinash Desai, Actinium's Chief Medical Officer, commented, "Improvements in overall survival have been difficult to achieve in relapsed or refractory AML patients, particularly for those with TP53 mutations. Additionally, as venetoclax-based treatments have become a standard of care, physicians need better treatment options to then manage the significant number of patients that do not respond, stop treatment due to toxicities or relapse and who have a median overall survival of just 2.4 months. We are incredibly excited by these data that clearly show Actimab-A's potential to improve patient outcomes when combined with CLAG-M. Further, it shows how Actimab-A can be utilized in combination with other therapies and add potency, which supports our strategy to establish it as a backbone therapy for AML with other therapies. With our recommended phase 2 dose finalized and these strong rates of MRD negativity and overall survival, we look forward to providing updates on our development and regulatory strategy as we work to bring this important combination to patients."
Title: Lintuzumab-Ac225 with Combination with Intensive Chemotherapy Yields High Response Rate and MRD Negativity in R/R AML with Adverse Features
Abstract Number: 65
Session Name: 616. Acute Myeloid Leukemias: Investigational Therapies, Excluding Transplantation and Cellular Immunotherapies: Relapsed/Refractory AML
Date: Saturday, December 10, 2022
Time: 10:30 AM CT
Location: Ernest M. Morial Convention Center, Room 220-222
References:
- Maiti et al. Outcomes of relapsed or refractory acute myeloid leukemia after front-line hypomethylating agent and venetoclax regimens. Hematoligica 2021 Mar 1; 894-898
- Ganzel et al. Very poor long-term survival in past and more recent studies for relapsed AML patients: The ECOG-ACRIN experience. American Journal of Hematology. 2018 Aug; 93(8): 1074–1081
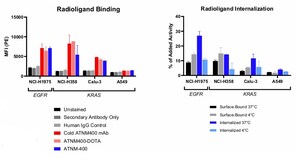
Actinium Pharmaceuticals, Inc. is a clinical-stage biopharmaceutical company developing targeted radiotherapies to deliver cancer-killing radiation with cellular level precision to treat patients with high unmet needs. Actinium's clinical pipeline is led by radiotherapies that are being applied to targeted conditioning, which is intended to selectively deplete a patient's disease or cancer cells and certain immune cells prior to a bone marrow transplant (BMT), gene therapy or adoptive cell therapy, such as CAR-T, to enable engraftment of these transplanted cells with minimal toxicities. Our lead product candidate, I-131 apamistamab (Iomab-B) has been studied in over four hundred patients, including the pivotal Phase 3 Study of Iomab-B in Elderly Relapsed or Refractory Acute Myeloid Leukemia (SIERRA) trial for BMT conditioning. Topline data from the SIERRA trial was positive with the study meeting its primary endpoint with a high statistical significance (p<0.0001). Additional data from the SIERRA trial is expected to be presented by year-end. Iomab-ACT, low dose I-131 apamistamab, is being studied as a targeted conditioning agent in a Phase 1 study with a CD19 CAR T-cell Therapy with Memorial Sloan Kettering Cancer Center with NIH funding. Actimab-A, our second most advanced product candidate has been studied in approximately 150 patients with Acute Myeloid Leukemia or AML, including in ongoing combination trials with the chemotherapy regimen CLAG-M and with venetoclax, a targeted therapy. Actimab-A or lintuzumab-Ac225 is an Actinium-225 based antibody radiation conjugate targeting CD33, a validated target in AML. Actinium is a pioneer and leader in the field of Actinium-225 alpha therapies with an industry leading technology platform comprising over 190 patents and patent applications including methods of producing the radioisotope AC-225. Our technology and expertise have enabled collaborative research partnerships with Astellas Pharma, Inc. for solid tumor theranostics, with AVEO Oncology Inc. to create an Actinium-225 HER3 targeting radiotherapy for solid tumors, and with EpicentRx, Inc. to create targeted radiotherapy combinations with their novel, clinical stage small molecule CD47-SIRPα inhibitor. More information is available on Actinium's website: https://www.actiniumpharma.com/.
This press release may contain projections or other "forward-looking statements" within the meaning of the "safe-harbor" provisions of the private securities litigation reform act of 1995 regarding future events or the future financial performance of the Company which the Company undertakes no obligation to update. These statements are based on management's current expectations and are subject to risks and uncertainties that may cause actual results to differ materially from the anticipated or estimated future results, including the risks and uncertainties associated with preliminary study results varying from final results, estimates of potential markets for drugs under development, clinical trials, actions by the FDA and other governmental agencies, regulatory clearances, responses to regulatory matters, the market demand for and acceptance of Actinium's products and services, performance of clinical research organizations and other risks detailed from time to time in Actinium's filings with the Securities and Exchange Commission (the "SEC"), including without limitation its most recent annual report on form 10-K, subsequent quarterly reports on Forms 10-Q and Forms 8-K, each as amended and supplemented from time to time.
Investors:
Hans Vitzthum
LifeSci Advisors, LLC
[email protected]
(617) 430-7578
SOURCE Actinium Pharmaceuticals, Inc.






Share this article