
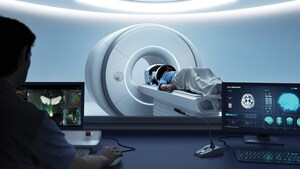
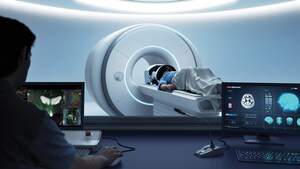
Exablate Prostate system uses Focused Ultrasound guided by MRI
HAIFA, Israel and MIAMI, Fla., Dec. 1, 2021 /PRNewswire/ -- Insightec, a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy, announced today that the Exablate Prostate system received FDA 510K clearance for treating prostate tissue with high intensity Focused Ultrasound.
The Exablate Prostate system uses sound waves to ablate, or destroy, targeted tissue in the prostate. The treatment is performed under Magnetic Resonance Imaging (MRI) guidance for high resolution visualization of the patient's anatomy for precise targeting and real-time temperature monitoring. The treatment does not require incisions, is performed in a single session, allowing patients to quickly return to normal activity with minimal complications.
"Exablate Focused Ultrasound has been shown to provide effective control of locally-confined prostate cancer in select patients based on 2-year biopsy outcomes," said Behfar Ehdaie, MD, MS, a urologic surgeon at Memorial Sloan Kettering Cancer Center, and trial PI. "Precision ablation together with MR imaging and thermal feedback may enable men to consider a tissue preserving approach and defer or avoid radical therapy."
The Insightec-sponsored clinical trial demonstrated that Exablate Prostate resulted in minimal damage to adjacent structures, with low rates of impact on potency and continence.
"The results in our clinical trial patients undergoing focal therapy using Exablate have been truly impressive. I'm very excited to bring this new technology to more men with localized prostate cancer," said Dr. Geoffrey Sonn, Assistant Professor of Urology at Stanford University.
"Insightec remains focused on achieving strategic milestones and expanding into indications beyond Neurosurgery such as approval for prostate tissue ablation," said Maurice R. Ferré MD, Insightec's CEO and Chairman of the Board. "Incisionless surgery is transforming patient care with a surgical option that is considerably less invasive."
About Insightec
Insightec is a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy. The company's Exablate Neuro platform focuses sound waves, safely guided by MRI, to provide tremor treatment to patients with medication-refractory Essential Tremor and Tremor-dominant Parkinson's Disease. Research for future applications in the neuroscience space is underway in partnership with leading academic and medical institutions. Insightec is headquartered in Haifa, Israel, and Miami, with offices in Dallas, Shanghai, and Tokyo.
Follow us on Facebook, LinkedIn and Twitter or visit www.insightec.com for more information.
Forward-looking Statements:
This document contains forward-looking statements regarding, among other things, plans, expectations, and future events. In some cases, forward-looking statements can be identified by the following words: "may," "can," "will," "could," "would," "should," "expect," "intend," "plan," "anticipate," "believe," "estimate," "predict," "project," "potential," "promise," "continue," "ongoing," or the negative of these terms. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that could cause actual results to differ materially from what is expressed or implied by the statements. Any forward-looking statement is based on information available to Insightec as of the date of the statement. All written or oral forward-looking statements attributable to Insightec are qualified by this caution. Insightec does not undertake any obligation to update or revise any forward-looking statement to reflect any change in circumstances or in Insightec's expectations.
"Exablate," and "Exablate Neuro," as well as the "INSIGHTEC" logo, whether standing alone or in connection with the word "Insightec", are protected trademarks of Insightec.
Insightec Media Contact:
G&S Business Communications for Insightec
Aaliyah Alexander
+1 917.595.3064
[email protected]
SOURCE Insightec






Share this article