
Latest Clearance Completes MedCAD's Lower Extremity Offering
DALLAS, April 8, 2026 /PRNewswire/ -- Dallas-based MedCAD has received FDA 510(k) clearance of its AccuStride® lower leg tibia and fibula system, making these patient-specific precision devices available to surgeons. The unique design of the custom-made planning and surgical guides will enable orthopedic surgeons to correct multiple related pathologies and osteotomies in a single procedure.
The company recently received 510(k) FDA clearance for its foot and ankle guides and planning system in March 2025. In November 2025 MedCAD received 510(k) FDA clearance for its fixation plates, part of its patient-specific 3D-printed solution available to surgeons as a complete foot and ankle (F&A) system. The newest 510(k) FDA clearance now completes the patent-pending AccuStride® system offering for lower extremities: tibia/fibula, foot and ankle.
"MedCAD's newly announced, patient-matched surgical guides are unlike anything else available for surgeons who routinely perform complex or revision lower leg region cases," said Nancy Hairston, CEO and president of MedCAD. "Interest from leading orthopedic specialists eager to use our solutions for multiple pathologies is very promising, and we expect these custom 3-D printed devices to reduce the frequency and duration of surgeries, and to deliver high quality, durable outcomes."
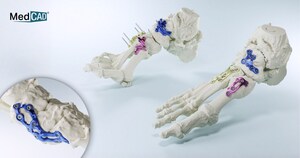
The AccuStride™ System includes surgical guides, plates and anatomical models. Components are constructed of UV-curable acrylate polymers or titanium alloy materials and can be delivered in as few as five days after surgeon design approval. The system is intended for use in patients 12 years and older.
System guides provide tactile placement of specific surgical procedural steps derived from the patient imaging data and the surgeon approved plan. The titanium guides are compatible with FDA-cleared surgical cutting/drilling instruments, and the physician selects the appropriate instruments to interface with each type of surgical device. The system's anatomical models provide visual representation of a specific patient's anatomy derived from the patient imaging data and the surgeon approved plan.
"The goal of MedCAD's portfolio of patient-matched product solutions is to provide surgeons with customizable options that are as anatomically unique as their patients," said Hairston. "MedCAD has been building a strong reputation for putting cutting-edge planning technology into the hands of surgeons so that they can deliver life-changing outcomes for the people they heal. With AccuStride, we recognized that when one part of a lower leg is experiencing problems, significant issues can show up in other areas as well, and with our advanced, wholistic approach, the outcomes are unmatched, and the possibilities are virtually endless."
MedCAD is also a leading manufacturer of patient-specific, custom-designed cranial and maxillofacial reconstruction implants, including virtual planning and surgical devices.
About MedCAD
MedCAD is a Dallas-based, privately held medical technology company built on an innovative approach to the design and production of patient-matched medical devices. Harnessing precise imaging, surgical experience, and advanced design and manufacturing technologies, MedCAD creates personalized, patient-matched medical devices and surgical plans for cranial defects, oral surgery, CMF trauma, and reconstructive surgical procedures. The approach is 100% patient-customized, with every device and every procedure planned and manufactured in-house in cooperation with a patient's surgeon. By minimizing surgical complexity and procedure time, MedCAD technology enables superior patient outcomes throughout intervention, rehabilitation, and recovery.
SOURCE MedCAD





Share this article