
Mylan Taking Immediate Action to Further Enhance Access to EpiPen® (Epinephrine Injection, USP) Auto-Injector
Mylan doubling the eligibility for patient assistance, effectively eliminating out-of-pocket expense for uninsured and under-insured patients
Company to cover up to $300 of out-of-pocket cost at pharmacy; reducing patient cost by 50% off Mylan list price
Mylan also to open pathway to provide EpiPen® Auto-Injector directly to patients
HERTFORDSHIRE, England and PITTSBURGH, Aug. 25, 2016 /PRNewswire/ -- Mylan N.V. (NASDAQ, TASE: MYL) today announced it is taking immediate action to further enhance access to EpiPen® (epinephrine injection, USP) Auto-Injector by expanding already existing programs in recognition of those patients who are facing the burden of higher out-of-pocket costs. The company is reducing the patient cost of EpiPen® Auto-Injector through the use of a savings card which will cover up to $300 for their EpiPen 2-Pak®. For patients who were previously paying the full amount of the company's list price for EpiPen®, this effectively reduces their out-of-pocket cost exposure by 50%. Mylan also is doubling the eligibility for its patient assistance program, which will eliminate out-of-pocket costs for uninsured and under-insured patients and families as well.
Mylan CEO Heather Bresch said, "We have been a long-term, committed partner to the allergy community and are taking immediate action to help ensure that everyone who needs an EpiPen® Auto-Injector gets one. We recognize the significant burden on patients from continued, rising insurance premiums and being forced increasingly to pay the full list price for medicines at the pharmacy counter. Patients deserve increased price transparency and affordable care, particularly as the system shifts significant costs to them. However, price is only one part of the problem that we are addressing with today's actions. All involved must also take steps to help meaningfully address the U.S. healthcare crisis, and we are committed to do our part to drive change in collaboration with policymakers, payors, patients and healthcare professionals."
Mylan has worked to help patients with commercial insurance pay as little as $0 for EpiPen® Auto-Injector using the My EpiPen Savings Card®. In 2015, this resulted in nearly 80% of these patients paying nothing out of pocket for their EpiPen® Auto-Injector. However, as the health insurance environment has evolved, driven by the implementation of the Affordable Care Act, patients and families enrolled in high deductible health insurance plans, who are uninsured, or who pay cash at the pharmacy, have faced higher costs for their medicine.
Mylan is taking the following immediate actions to help further address the needs of patients and families:
- For patients in health plans who face higher out-of-pocket costs, the company is providing immediate relief by offering a savings card for up to $300. This will effectively reduce by 50% the cost exposure for patients who would have otherwise paid the full list price for EpiPen® Auto-Injector.
- Mylan also is doubling eligibility for our patient assistance program to 400% of the federal poverty level. This means a family of four making up to $97,200 would pay nothing out of pocket for their EpiPen® Auto-Injector.
- Further, Mylan will continue to offer the EpiPen4Schools® program. The program, launched in August 2012, has provided more than 700,000 free epinephrine auto-injectors and educational resources to more than 65,000 schools nationwide to help them be prepared for anaphylaxis events among students.
- Mylan also is opening a pathway so that patients can order EpiPen® Auto-Injector directly from the company, thereby reducing the cost.
These programs will apply to EpiPen® and EpiPen Jr® (epinephrine injection, USP) Auto-Injectors.
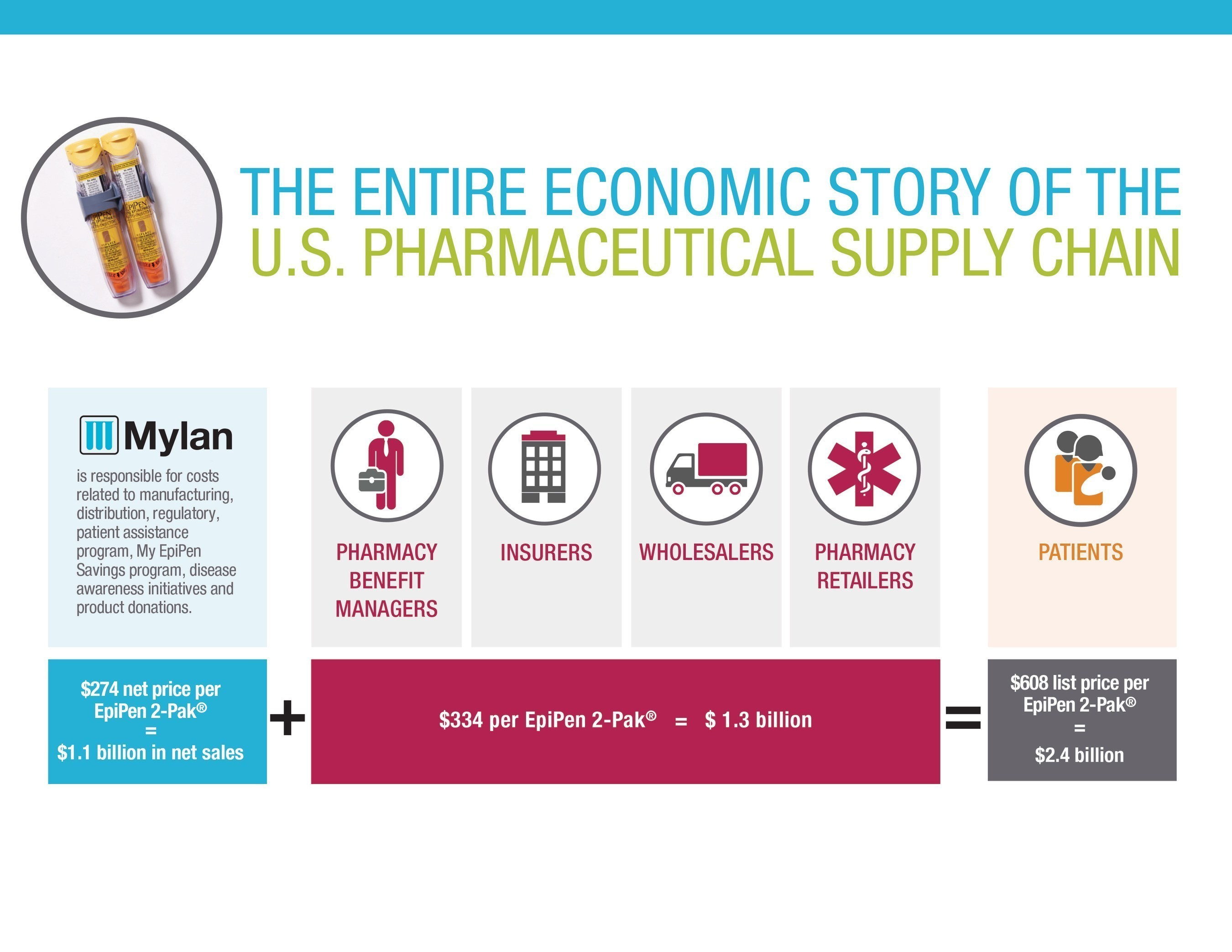
In the interest of fostering greater price transparency in the healthcare system, Mylan is providing the entire economic story of the EpiPen® supply chain on our website.
About EpiPen® (epinephrine injection, USP) Auto-Injector
EpiPen® (epinephrine injection, USP) and EpiPen Jr® (epinephrine injection, USP) Auto-Injectors are used for the emergency treatment of life-threatening allergic reactions. Each EpiPen 2-Pak® and EpiPen Jr 2-Pak® contains two single auto-injectors, instructions for use and a training device, with no drug product or needle, to help patients become familiar with the administration technique. EpiPen® Auto-Injector should be administered immediately at the first sign of an anaphylactic reaction. EpiPen® Auto-Injector is not a substitute for emergency medical treatment. Patients should seek emergency medical attention immediately following administration. For more information about EpiPen® Auto-Injector, visit epipen.com.
About Mylan
Mylan is a global pharmaceutical company committed to setting new standards in healthcare. Working together around the world to provide 7 billion people access to high quality medicine, we innovate to satisfy unmet needs; make reliability and service excellence a habit; do what's right, not what's easy; and impact the future through passionate global leadership. We offer a growing portfolio of more than 2,700 generic and branded pharmaceuticals, including antiretroviral therapies on which approximately 50% of people being treated for HIV/AIDS in the developing world depend. We market our products in more than 165 countries and territories. Our global R&D and manufacturing platform includes more than 50 facilities, and we are one of the world's largest producers of active pharmaceutical ingredients. Every member of our more than 40,000-strong workforce is dedicated to creating better health for a better world, one person at a time. Learn more at mylan.com
Indications
EpiPen® (epinephrine injection, USP) 0.3 mg and EpiPen Jr® (epinephrine injection, USP) 0.15 mg Auto-Injectors are for the emergency treatment of life-threatening allergic reactions (anaphylaxis) caused by allergens, exercise, or unknown triggers; and for people who are at increased risk for these reactions. EpiPen® and EpiPen Jr® are intended for immediate administration as emergency supportive therapy only. Seek immediate emergency medical help right away.
Important Safety Information
Use EpiPen® or EpiPen Jr® Auto-Injectors right away when you have an allergic emergency (anaphylaxis). Get emergency medical help right away. You may need further medical attention. Only a healthcare professional should give additional doses of epinephrine if you need more than two injections for a single anaphylactic episode. EpiPen® or EpiPen Jr® should only be injected into the middle of your outer thigh (upper leg), through clothing if necessary. Do not inject into your veins, buttocks, fingers, toes, hands or feet. Hold the leg of young children firmly in place before and during injection to prevent injuries. In case of accidental injection, please seek immediate medical treatment.
Rarely, patients who have used EpiPen® or EpiPen Jr® may develop an infection at the injection site within a few days. Some of these infections can be serious. Call your healthcare professional right away if you have any of the following at an injection site: redness that does not go away, swelling, tenderness, or the area feels warm to the touch.
Tell your healthcare professional about all of your medical conditions, especially if you have asthma, a history of depression, thyroid problems, Parkinson's disease, diabetes, high blood pressure or heart problems, have any other medical conditions, are pregnant or plan to become pregnant, or are breastfeeding or plan to breastfeed. Be sure to also tell your healthcare professional all the medicines you take, especially medicines for asthma. If you have certain medical conditions, or take certain medicines, your condition may get worse or you may have longer lasting side effects when you use EpiPen® or EpiPen Jr®.
Common side effects include fast, irregular or "pounding" heartbeat, sweating, nausea or vomiting, breathing problems, paleness, dizziness, weakness, shakiness, headache, feelings of over excitement, nervousness or anxiety. These side effects usually go away quickly if you lie down and rest. Tell your healthcare professional if you have any side effect that bothers you or that does not go away.
Please see the full Prescribing Information and Patient Information.
You are encouraged to report negative side effects of prescription drugs to the FDA.
Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
For additional information, please contact us at 800-395-3376.
EpiPen®, EpiPen Jr®, EpiPen 2-Pak®, EpiPen Jr 2-Pak®, EpiPen4Schools® and My EpiPen Savings Card® are registered trademarks owned by Mylan Inc.
© 2016 Mylan Specialty L.P. All rights reserved.
Photo - http://photos.prnewswire.com/prnh/20160825/401225-INFO
Photo - http://photos.prnewswire.com/prnh/20160825/401226-INFO
SOURCE Mylan N.V.


Share this article