
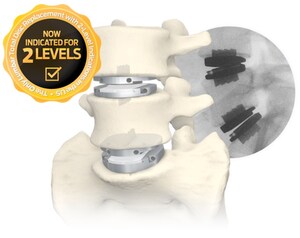
NEW YORK, May 2, 2019 /PRNewswire/ -- Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced findings of a new study first presented at the annual meeting of the International Society for the Advancement of Spinal Surgery (3-5 April 2019, Anaheim, CA). Jack E. Zigler, MD (Texas Health Center for Diagnostics and Surgery, Texas Back Institute, Plano, TX) reported results that "strongly support" the safety of Centinel Spine's prodisc® C Cervical Total Disc Replacement System. In the study of 504 consecutive patients, Zigler and colleagues found no secondary surgery requirements for device failure and a relatively low reoperation rate of 5.5%—including the learning curve for every participating surgeon. The study encompassed results, specifically reoperation rates, for all consecutive patients receiving a prodisc C over a fifteen-year timeframe at a single center.
"This is a complete data set on a large series of consecutive cases with a single implant. It also represents the early learning curve in cervical arthroplasty for all surgeons involved," commented Zigler on the significance of the study. "A very low reoperation rate and no instance of device breakage were two positive findings of the study," he continued.
This long-term study further supports multiple shorter-term multi-center prodisc C studies with comparable secondary operation rates. The single-center design of the study eliminated many surgical and data collection variables seen in multi-center analyses. It also addressed multi-center study weaknesses, such as learning curve effects, as prodisc C was the first arthroplasty device utilized by many of the surgeons in the study.
"As a result of our experiences with prodisc C and other systems, total disc replacement has evolved to become our primary choice for managing patients with degenerative disc disease. The prodisc C safety profile confirmed through this study validates our approach to patient care—patients need to know they have safe and effective alternatives to fusion," says Zigler.
Centinel Spine's prodisc device is the most studied and clinically-proven Total Disc Replacement system in the world. The prodisc design has been validated with over 125,000 device implantations and over 400 published studies reporting on over 14,000 patient experiences. This study added 504 more patient experiences to the published body of evidence supporting the safety and efficacy of prodisc C.
About Centinel Spine, LLC
Centinel Spine®, LLC is the largest privately-held spine company focused on anterior column reconstruction. The company offers a continuum of trusted, brand-name motion-preserving and fusion solutions backed by over 30 years of clinical success—providing the most robust and clinically-proven Total Disc Replacement and Integrated Interbody™ portfolios in the world.
The company began operations in 2008 through the merger-acquisition of two pioneering medical device companies—Raymedica, LLC and Surgicraft, LTD. UK-based Surgicraft launched the first Stand-Alone/No Profile® anterior lumbar interbody fusion device in the world in 1988, which was the basis for future generations of the market-leading Integrated Interbody™ technology platform known today as STALIF®. Today, Centinel Spine still embraces the pioneering culture developed at both originating companies and continues its corporate mission to become the worldwide leading company addressing spinal disease anteriorly with the widest breadth & depth of technology platforms.
The company recently acquired the prodisc® Total Disc Replacement Technology Platform—the most extensive cervical and lumbar motion-preserving reconstruction portfolio available today. With the addition of prodisc, Centinel Spine stands alone as the only company with comprehensive motion-preserving and fusion solutions for both cervical and lumbar anterior column reconstruction.
Centinel Spine derived its name from the "Sentinel Sign", the radiographic confirmation of a successful fusion anterior to the interbody device.
For more information, please visit the company's website at www.CentinelSpine.com or contact:
Varun Gandhi
SVP, Corporate Finance & Strategic Planning
900 Airport Road, Suite 3B
West Chester, PA 19380
Phone: 484-887-8871
Email: [email protected]
SOURCE Centinel Spine, LLC




Share this article