
Oncolytics Biotech® Inc. Reports Additional Data from Randomized Phase II Study of REOLYSIN® in Non-Small Cell Lung Cancer
--Study Demonstrates Statistically Significant Improvement in Progression Free Survival in Female Patients with Adenocarcinoma--
CALGARY, Aug. 10, 2016 /PRNewswire/ -- Oncolytics Biotech Inc. ("Oncolytics" or the "Company") (TSX: ONC) (OTCQX: ONCYF) (FRA: ONY) today announced additional data from IND 211, a randomized, Phase II clinical study of REOLYSIN® in patients with non-small cell lung cancer ("NSCLC"). The study enrolled patients with both non-squamous (adenocarcinoma) and squamous cell histology. Those with adenocarcinoma (n=75) were treated with REOLYSIN® in combination with pemetrexed in the test arm versus pemetrexed alone in the control arm. Those with squamous cell histology (n=76) were treated with REOLYSIN® in combination with docetaxel in the test arm versus docetaxel alone in the control arm. The study's primary objective was progression free survival ("PFS"). Its secondary objectives included overall survival ("OS"), safety, and measurement of biomarkers that may be predictive of response.
Results in Patients with Adenocarcinoma by Patient Gender
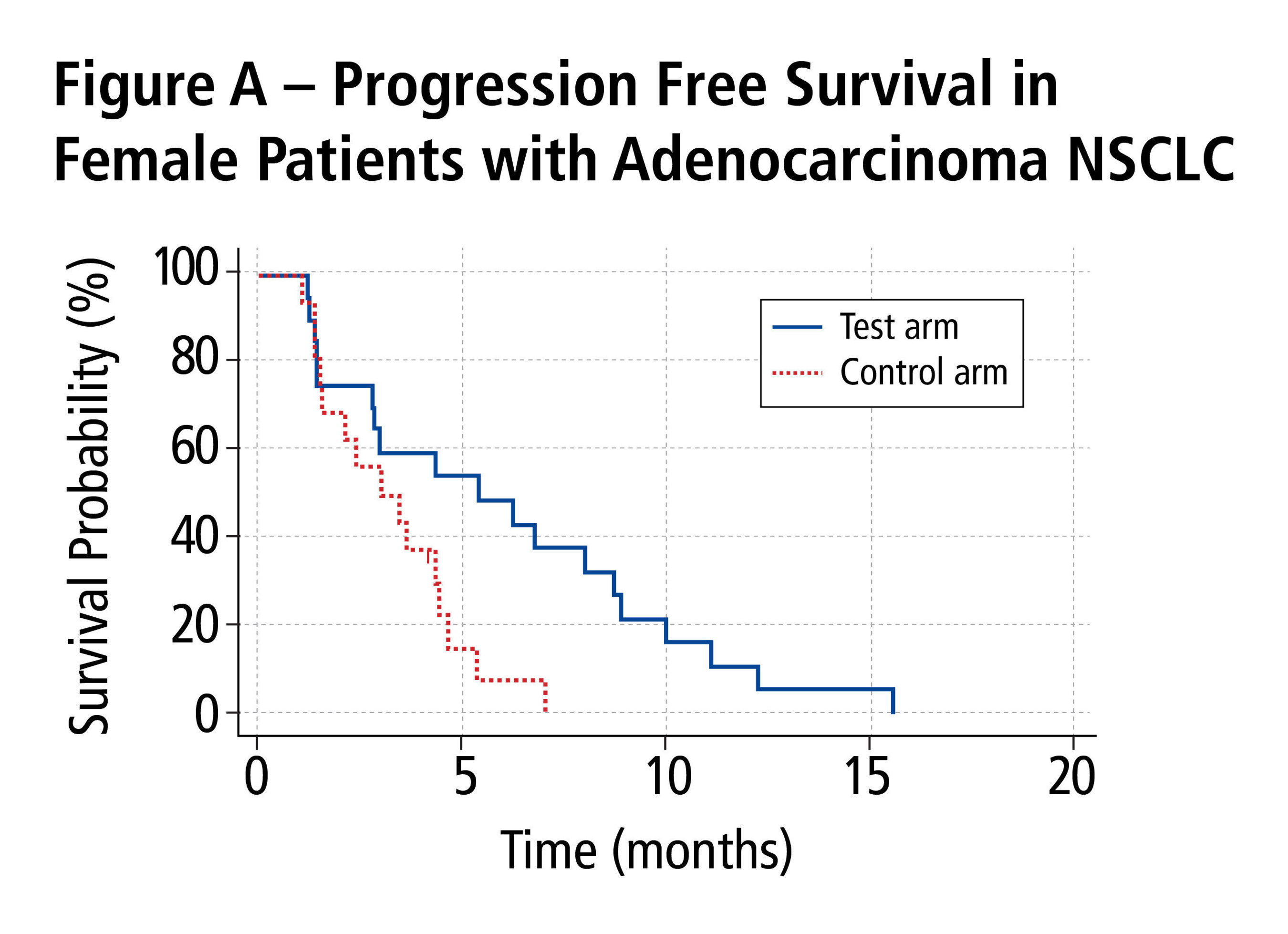
An analysis was performed of survival outcomes for patients with adenocarcinoma by gender. The PFS was statistically significantly better for female patients in the test arm (n=20) than for those in the control arm (n=16); median PFS was 5.39 months compared with 3.02 months, respectively (p=0.0201) (Figure A). The evolving OS showed a strong trend towards survival benefit for female patients in the test arm (n=20, six of whom remained alive at the time of the analysis) over those in the control arm (n=16, three of whom remained alive at the time of the analysis); median OS was 10.68 months compared with 7.59 months, respectively (p=0.145) (Figure B). By contrast, no PFS or OS benefit was noted for the male patients with adenocarcinoma.
"We are excited to see a statistically significant improvement in progression free survival for female patients with adenocarcinoma of the lung," said Dr. Brad Thompson, President and CEO of Oncolytics. "There is a significant unmet need for new therapies for non-small cell lung cancer patients."
Results in Patients with Adenocarcinoma by Patient Genetic Status
All patients were tested for biomarkers including those that are associated with the replication of the reovirus (namely, EGFR, Hras, Kras, Nras, Braf and/or p53 mutations) (the "target biomarkers"). Patients treated with REOLYSIN® with one or more target biomarkers had a greater PFS (p=0.039) and OS (p=0.031) than patients treated with REOLYSIN® without any of these biomarkers. The presence of these biomarkers may account, at least in part, for the difference between the survival outcomes for male and female patients; target biomarkers were present in a higher proportion of the female patients in the study than the male patients (66.7% versus 43.6%). As a result, pre-screening for target biomarkers in patients with adenocarcinoma of the lung is warranted.
Results in Patients with Squamous Cell Histology
The overall OS for patients with squamous cell histology continues to evolve. Target biomarkers were present in a smaller proportion of the overall patients with squamous cell histology (34.2% versus 54.7% of those with adenocarcinoma); therefore a larger patient population would be required to make statistical conclusions about the role of biomarkers in predicting response.
Next Steps
Based on the findings today announced from IND 211, the Company intends to include preselection of patients using genetic screening in future study protocols.
About IND 211
IND 211 is an open-label, randomized, non-blinded two sided (adenocarcinoma and squamous cell carcinoma) Phase II study of intravenously administered REOLYSIN® in patients with advanced or metastatic non-small cell lung cancer. Patients with squamous cell histology were treated with either REOLYSIN® given in combination with docetaxel (test arm) or docetaxel alone (control arm). Patients with adenocarcinoma were treated with either REOLYSIN® given in combination with pemetrexed (test arm) versus pemetrexed alone (control arm). After a patient safety run-in, a total of approximately 150 response-evaluable patients were enrolled. Preliminary data from this study were reported in May 2016. The study was sponsored by the Canadian Cancer Trials Group ("CCTG") at Queen's University in Kingston, Ontario, formerly known as the National Cancer Institute of Canada ("NCIC") Clinical Trials Group.
About Non-Small Cell Lung Cancer
The American Cancer Society ("ACS") estimates that approximately 224,390 Americans will be diagnosed with lung cancer in 2016 and that 158,080 of them will die from the disease. Approximately 80% to 85% of lung cancer cases are non-small cell lung cancer. The ACS identifies multiple subtypes of non-small cell lung cancer based on the types of cells where they originate – adenocarcinoma accounts for about 40% of lung cancers and occurs more frequently in women than men; squamous cell carcinoma accounts for 25% to 30% of lung cancers; and large cell carcinoma accounts for 10% to 15% of lung cancers.
About Oncolytics Biotech Inc.
Oncolytics is a Calgary-based biotechnology company focused on the development of oncolytic viruses as potential cancer therapeutics. Oncolytics' clinical program includes a variety of later-stage, randomized human trials in various indications using REOLYSIN®, its proprietary formulation of the human reovirus. For further information about Oncolytics, please visit: www.oncolyticsbiotech.com.
This press release contains forward-looking statements, within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements, including the Company's expectations related to the randomized Phase II study in patients with non-small cell lung cancer, future trials in this indication, and the Company's belief as to the potential of REOLYSIN® as a cancer therapeutic, involve known and unknown risks and uncertainties, which could cause the Company's actual results to differ materially from those in the forward-looking statements. Such risks and uncertainties include, among others, the availability of funds and resources to pursue research and development projects, the efficacy of REOLYSIN® as a cancer treatment, the tolerability of REOLYSIN® outside a controlled test, the success and timely completion of clinical studies and trials, the Company's ability to successfully commercialize REOLYSIN®, uncertainties related to the research, development and manufacturing of pharmaceuticals, changes in technology, general changes to the economic environment and uncertainties related to the regulatory process. Investors should consult the Company's quarterly and annual filings with the Canadian and U.S. securities commissions for additional information on risks and uncertainties relating to the forward-looking statements. Investors are cautioned against placing undue reliance on forward-looking statements. The Company does not undertake to update these forward-looking statements, except as required by applicable laws.
For further information: NATIONAL Equicom, Nick Hurst, 320 Front Street W, Suite 1600, Toronto, Ontario, M5V 3B6, Tel: 416.586.1942, [email protected]; NATIONAL Equicom, Michael Moore, San Diego, CA, Tel: 858.886.7813, [email protected]; Dian Griesel, Inc., Susan Forman, 335 West 38th Street, 3rd Floor, New York, NY, 10018, Tel: 212.825.3210, [email protected]
Photo - http://photos.prnewswire.com/prnh/20160810/397063
Photo - http://photos.prnewswire.com/prnh/20160810/397064
SOURCE Oncolytics Biotech Inc.


Share this article