
Long-term partnership secures high-quality active pharmaceutical ingredient supply for Lasix® ONYU and future pipeline products
BURLINGTON, Mass. and VICENZA, Italy, April 15, 2026 /PRNewswire/ -- SQ Innovation, the developer of Lasix® ONYU (furosemide injection), and FIS Fabbrica Italiana Sintetici S.p.A. today announced a strategic supply agreement to secure the long-term availability of pharmaceutical-grade furosemide for SQ Innovation's current and pipeline products across the United States, Europe, and global markets. FIS, a premier Italian manufacturer and one of Europe's foremost developers and producers of active pharmaceutical ingredients (APIs), will serve as a key supply partner supporting SQ Innovation's mission to deliver reliable, high-quality therapies to patients worldwide.
Under the terms of the agreement, FIS will provide SQ Innovation with pharmaceutical-grade furosemide API to support the continued production of Lasix ONYU, a drug-device combination product approved by the US Food and Drug Administration in 2025 for at-home treatment of edema in adult patients with heart failure. The partnership also supports SQ Innovation's development pipeline and planned geographic expansion. The agreement includes certain exclusivity provisions.
"Securing a reliable supply of high-quality furosemide API is critical to our mission of providing innovative heart failure treatment options that aim to provide an alternative to hospitalizations," said Pieter Muntendam, MD, Founder, President and CEO at SQ Innovation. "FIS's reputation for excellence in API manufacturing and their commitment to quality align with our standards and our growth objectives."
"We are proud to partner with SQ Innovation in advancing transformative solutions for heart failure treatment,' said Daniele Piergentili, CEO and Chairman at FIS. 'By combining our advanced manufacturing expertise with rigorous quality and regulatory standards, we can provide the strong, reliable supply foundation for this proven ingredient for innovative products to the benefit of patients worldwide."
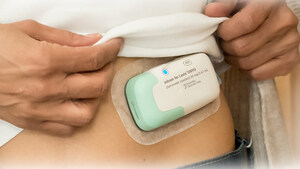
About Lasix ONYU
Lasix® ONYU is a drug-device combination that was approved by the U.S. Food and Drug Administration on October 7, 2025 for the treatment of edema in adult patients with chronic heart failure. Lasix ONYU is available for ordering through Cardinal Health's Specialty Pharmaceutical Distribution (SPD) and Cencora's Specialty Distribution (formerly AmerisourceBergen). The pharmaceutical component of Lasix ONYU is a novel, high-concentration formulation of the diuretic furosemide, at 30 mg/mL. It comes in a pre-filled glass cartridge containing 80 mg of furosemide in 2.67 mL. The Lasix ONYU Infusor consists of two main parts: the Reusable Unit and the Disposable Unit. The Reusable Unit is an electromechanical device that contains the battery, motor, and electronic components necessary for operation and safety functions. It can be used up to 48 times before it can be recycled. The Disposable Unit is a sterile, single-use plastic component that holds the drug cartridge. It includes a micropiston pump, fluid path, needle insertion and retraction mechanism, and a 29-gauge needle. After placement on the abdomen, the needle penetrates the skin when the device is activated. The Lasix ONYU Infusor slowly administers 80 mg furosemide over a period of five hours. This method results in significant diuresis similar to IV, but in a more controlled manner. This avoids the brief, intense diuretic effect that occurs with rapid IV infusion or injection. The advanced two component design offers benefits for patients, healthcare providers, payers, and the environment. For Important Safety Information, Prescribing Information and Instructions for Use, visit www.lasix-onyu.com.
INDICATION
Lasix® ONYU is a loop diuretic indicated for the treatment of edema in adult patients with chronic heart failure.
IMPORTANT SAFETY INFORMATION
Before using Lasix® ONYU, read the Instructions for Use and tell your healthcare provider about all your medical conditions, including if you are allergic to furosemide or any of the ingredients in Lasix ONYU, have trouble urinating, or if you are allergic to medical adhesives.
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines.
Warning: Only use the Lasix ONYU Prefilled Cartridge with the Lasix ONYU Infusor. Do not use insulin cartridges or other medicine cartridges in the Lasix ONYU Infusor. Doing so could cause severe injury.
What are the possible side effects of Lasix ONYU?
Dehydration: Lasix ONYU is a diuretic that can make you lose a lot of fluid and with it electrolytes. You may get a dry mouth, have increased thirst, get muscle pains or cramps, have reduced urine output or your urine may be more yellow than normal, you may get a headache, get dry skin, or have nausea or vomiting. Your healthcare provider may check your electrolytes while receiving Lasix ONYU.
Low Blood Pressure: Lasix ONYU may lower your blood pressure temporarily. You may feel lightheaded or dizzy. This usually happens when you stand. Getting up slowly may help.
High Blood Sugar: Lasix ONYU may increase blood sugar (glucose) levels if you have diabetes mellitus.
Loss of Hearing: Lasix ONYU can cause ringing in your ears. If so, tell your healthcare provider.
Risk of Sunburn: Your skin may be more sensitive to sunlight while taking Lasix ONYU.
Infusion Site Reactions: Lasix ONYU can cause infusion site pain, bruising and temporary swelling or redness at the site of the Infusor.
Incomplete Dosing: Make sure the Infusor does not get wet during use. Also limit your physical activities. Some movements or when it gets wet may stop the infusion and you may not get all the medication.
These are not all the possible side effects of Lasix ONYU. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
Please see the full Prescribing Information and Instructions for Use.
About SQ Innovation
SQ Innovation, Inc. is a privately held Swiss biopharmaceutical company with offices in Zug, Switzerland, Burlington, MA, USA, and Rotterdam, The Netherlands. The company was founded to develop and commercialize innovative, cost-effective therapies for subcutaneous delivery, enabling at-home treatment for conditions that are usually managed during hospitalizations. SQ Innovation has developed a novel drug-device combination for treating edema in adult patients with chronic heart failure — a condition typically requiring intravenous administration of diuretics in a hospital setting. Lasix ONYU was developed with consideration for patients, payors, healthcare providers, and environmental impact. Lasix ONYU received approval from the US Food and Drug Administration on October 7, 2025. For more information about Lasix ONYU, including important safety information and the full prescribing information, please visit www.lasix-onyu.com.
For Media Inquiries: [email protected]
ABOUT FIS
Founded in 1957 in Montecchio Maggiore (Vicenza, Italy) and now controlled by Bain Capital Private Equity, FIS (Fabbrica Italiana Sintetici S.p.A.) is Italy's leader and one of the top players in Europe in the development and production of Active Pharmaceutical Ingredients (APIs) for the global pharmaceutical industry, standing out as an international benchmark in the CDMO (Contract Development and Manufacturing Organization) sector. FIS operates through three plants in Italy (Montecchio Maggiore, Termoli and Lonigo), commercial offices in North America and Japan, and operating sites in China and India. Today, it has over 2,300 employees, of whom approximately 270 dedicated to R&D activities. As of April 2024, FIS has been headed by Chief Executive Officer and Chairman Daniele Piergentili.
Press Contact
Micaela Colamasi, Head of Corporate & Product Communication
Ph. 338 71 88 939 | Email. [email protected]
SOURCE SQ Innovation







Share this article