
Updated Guidelines Significantly Expand Patient Access to Centinel Spine's prodisc® L Lumbar Total Disc Replacement
-- Patient access expansion is a result of updated medical necessity guidelines by National Imaging Associates (NIA), a Magellan Health Company.
-- Based on updates, nine major commercial payers will begin covering Total Disc Replacement (TDR) procedures with prodisc® L, representing an additional 9.2 million covered lives.
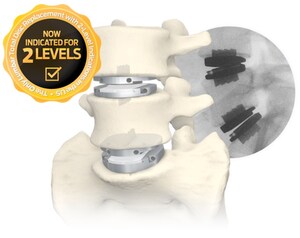
-- prodisc L has been clinically utilized across the world for over 20 years and is the only total disc replacement system in the U.S. approved for two-level use in the lumbar spine.
NEW YORK, July 22, 2020 /PRNewswire/ -- Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced a significant expansion to patient access for the prodisc® L Lumbar Total Disc Replacement (TDR) system. This represents an additional 9.2 million covered lives and is a result of an update to medical necessity guidelines by National Imaging Associates (NIA), a Magellan Health Company, to now include a positive coverage policy recommendation for lumbar TDR.
NIA Magellan provides third-party prior authorization services to U.S. insurers. Nine major commercial payers that follow NIA Magellan's clinical coverage guidelines have now started to cover lumbar TDR, including BCBS of Florida & S. Carolina, Harvard Pilgrim and Tufts, among others. Centinel Spine remains the only company in the world with an FDA-approved lumbar TDR device that has been clinically reviewed and found safe and effective for two-level use.
"This recent announcement of expanded coverage for lumbar TDR demonstrates the important progress being made in this area," stated Centinel Spine CEO Steve Murray. "We are encouraged that NIA Magellan recognizes the large body of clinical evidence that demonstrates lumbar TDR to be a safe and effective procedure for select patients," continued Murray. "This coverage expansion represents an excellent opportunity for surgeons to provide more patients with a broader spectrum of treatment options and ensure the delivery of the best spine care possible."
Centinel Spine stands alone as the only company with FDA-approved cervical and lumbar total disc replacement devices. New developments to the family of products include the recent FDA approval of two-level indications for the prodisc L Lumbar Total Disc Replacement system and a recently-initiated clinical trial comparing the prodisc C Vivo and prodisc C SK devices with an approved cervical TDR product as a control, in order to validate their safety and effectiveness in an FDA IDE study.
About Centinel Spine, LLC
Centinel Spine®, LLC is the largest privately-held spine company focused on anterior column reconstruction. The company offers a continuum of trusted, brand-name, motion-preserving and fusion solutions backed by over 30 years of clinical success—providing the most robust and clinically-proven Total Disc Replacement and Integrated Interbody™ portfolios in the world.
The company began operations in 2008 through the merger-acquisition of two pioneering medical device companies—Raymedica, LLC and Surgicraft, LTD. In 1988, UK-based Surgicraft launched the first Stand-Alone/No Profile® anterior lumbar interbody fusion device in the world, which was the basis for future generations of the market-leading Integrated Interbody technology platform known today as STALIF®. Today, Centinel Spine still embraces the pioneering culture developed at both originating companies and continues its corporate mission to become the worldwide leading company addressing spinal disease anteriorly with the widest breadth and depth of technology platforms.
In December, 2017, the company acquired the prodisc® Total Disc Replacement Technology Platform—the most extensive cervical and lumbar motion-preserving reconstruction portfolio available today. With the addition of prodisc, Centinel Spine stands alone as the only company with comprehensive motion-preserving and fusion solutions for both cervical and lumbar anterior column reconstruction.
In June, 2019, the company entered into partnership with professional athlete Tiger Woods. Woods underwent spinal fusion surgery using Centinel Spine's STALIF M-Ti™ Anterior Lumbar Integrated Interbody fusion product in April 2017 to alleviate ongoing, debilitating pain in his back and legs.
Centinel Spine derived its name from the "Sentinel Sign", the radiographic confirmation of a successful fusion anterior to the interbody device.
For more information, please visit the company's website at www.CentinelSpine.com or contact:
Varun Gandhi
SVP, Corporate Finance & Strategic Planning
900 Airport Road, Suite 3B
West Chester, PA 19380
Phone: 484-887-8871
Email: [email protected]
SOURCE Centinel Spine, LLC





Share this article