
BD Receives Order from Dutch Ministry of Health for More than Nine Million 15-minute COVID-19 Antigen Tests
The first shipment of 1.2 million SARS-CoV-2 Assays for use on the BD Veritor™ Plus System to be delivered in The Netherlands by mid-November
THE HAGUE, NETHERLANDS and FRANKLIN LAKES, N.J., Nov. 2, 2020 /PRNewswire/ -- BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced the receipt of an order from the Dutch Ministry of Health for 9.2 million of its rapid, point-of-care, SARS-CoV-2 antigen tests for use on the BD Veritor™ Plus System, which detects SARS-CoV-2 in symptomatic patients in approximately 15 minutes. The first 1.2 million units will be delivered by mid-November.
This is the first government contract in Europe for this test, which was CE marked to the IVD Directive (98/79/EC) in September. The Dutch Ministry of Health increased its initial order by 8 million assays to improve testing capacity. BD expects the order to be fulfilled by June 2021.
"We're proud to be working with the Dutch Ministry of Health to expand rapid testing for SARS-CoV-2. This test will be critical to aid rapid diagnosis and decision making," says Fernand Goldblat, vice president and general manager, Integrated Diagnostic Solutions, Europe at BD. "This rapid game-changing test is the latest effort in BD's comprehensive response to address the world's critical health needs during the global pandemic."
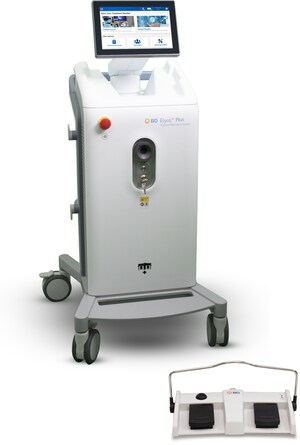
The BD Veritor™ Plus System is a portable, easy-to-use device that makes it an ideal solution for point-of-care settings. The BD Veritor™ Plus System is already in use across Europe to detect Influenza A+B, Respiratory Syncytial Virus (RSV), and Group A Strep. The SARS-CoV-2 assay has been available in the United States since July through an Emergency Use Authorization by the U.S. Food and Drug Administration (FDA).
For Product Enquiries and How to Order:
All BD SARS-CoV-2 diagnostic products have regulatory authorizations in the markets where they are sold. European customers interested in BD diagnostic solutions for SARS-CoV-2 should visit bd.com/VeritorSystem-EU or contact BD's local customer service.
About BD Veritor™ System for Rapid Detection of SARS-CoV-2 Assay
The BD Veritor™ Plus System for Rapid Detection of SARS-CoV-2 Assay has been CE marked to the IVD Directive (98/79/EC), but has not been cleared or approved by FDA. The test has been authorized by FDA under an Emergency Use Authorization (EUA) for use by authorized laboratories. The test has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens; and, the test is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner. The BD Veritor™ Plus System for Rapid Detection of SARS-CoV-2 Assay is not authorized for use by consumers or for at-home use.
About BD
BD is one of the largest global medical technology companies in the world and is advancing the world of health by improving medical discovery, diagnostics and the delivery of care. The company supports the heroes on the frontlines of health care by developing innovative technology, services and solutions that help advance both clinical therapy for patients and clinical process for health care providers. BD and its 65,000 employees have a passion and commitment to help enhance the safety and efficiency of clinicians' care delivery process, enable laboratory scientists to accurately detect disease and advance researchers' capabilities to develop the next generation of diagnostics and therapeutics. BD has a presence in virtually every country and partners with organizations around the world to address some of the most challenging global health issues. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase efficiencies, improve safety and expand access to health care.
For more information on BD, please visit bd.com.
Contacts:
|
|
Kristen Cardillo |
Kristen M. Stewart, CFA |
BD Public Relations |
BD Investor Relations |
201.847.5657 |
201.847.5378 |
Frankie Dundon |
|
For BD Europe |
|
SOURCE BD (Becton, Dickinson and Company)







Share this article