
CARsgen Therapeutics Joins The Alliance for Regenerative Medicine
SAN DIEGO, Calif., June 30, 2017 /PRNewswire/ --CARsgen Therapeutics (CARsgen), a global pioneer focused on developing chimeric antigen receptor T-cell (CAR-T cell) therapies for solid tumors, today announced it has become the active member of The Alliance for Regenerative Medicine (ARM) as the first Chinese company of advanced therapy.
Founded in 2009,The Alliance for Regenerative Medicine (ARM) is an international multi-stakeholder advocacy organization that promotes legislative, regulatory and reimbursement initiatives necessary to facilitate access to life-giving advances in regenerative medicine worldwide. ARM is the leading global advocacy organization to foster research, development, investment and commercialization of transformational treatments and cures. ARM also works to increase public understanding of the field and its potential to transform human healthcare, providing business development and investor outreach services to support the growth of its member companies and research organizations. Today, ARM has more than 250 members, ranging from leading global pharmaceutical such as Johnson & Johnson, Pfizer, and Novartis, leading biotech companies in advanced therapy such as Kite, Juno, Bluebird Bio, Lonza and PCT, to non-profit institutions, foundations and other stakeholders of global influence. All ARM members are committed to following the code of conduct set up by the Alliance and increase the availability of regenerative medicine to patients worldwide.
About CARsgen Therapeutics
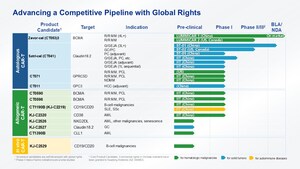
CARsgen Therapeutics is a rapidly growing bio-tech start-up with the original CAR-T therapy discovery expertise, competitive GMP manufacturing capability, and global clinical development experience. CARsgen commit to deliver the most advanced yet affordable cell therapies to patients with unmet medical needs. With broad discovery pipeline across solid tumor and hematology malignancies, CARsgen proceeds the clinical development with a clear focus in CAR-T therapy for solid tumors. Founded in Nov 2014, CARsgen raised $30M in early 2016 in Series B. Four of its pioneering CAR-T cell therapies, anti-GPC3 CAR-T for hepatocellular carcinoma (HCC), anti-GPC3 CAR-T for squamous lung cancer (SLC), cancer-specific anti-EGFR CAR-T for glioblastoma multiforme (GBM) and first-in-class anti-Claudin18.2-CAR-T for gastric and pancreatic cancer have all entered early clinical development.
For more information, please visit the website: www.carsgen.com
SOURCE CARsgen Therapeutics




Share this article