
SUNNYVALE, Calif., April 30, 2019 /PRNewswire/ -- EBR Systems, Inc., developer of the world's only wireless cardiac pacing system for heart failure, today announced that Dr. Mary Norine "Minnow" Walsh has become Co-Principal Investigator (Co-PI) of the SOLVE CRT clinical study. The SOLVE CRT (Stimulation of the Left Ventricular Endocardium for Cardiac Resynchronization Therapy) clinical study is a prospective randomized, double blinded pivotal trial intended to assess safety and efficacy of the WiSE (Wireless Stimulation Endocardially) pacing technology in support of U.S. Food and Drug Administration (FDA) approval. The study is expected to randomize 350 heart failure patients in the United States, Europe, and Australia who have failed to respond to, or are otherwise unable to receive, conventional cardiac resynchronization therapy (CRT).
Dr. Walsh, Medical Director of Heart Failure and Cardiac Transplantation at St. Vincent Heart Center in Indianapolis, will join Jagmeet P. Singh, M.D., Ph.D., of Massachusetts General Hospital in Boston to direct this landmark study. Dr. Walsh is a named author of over 100 publications, past president of the American College of Cardiology (ACC), and a former member of the Heart Failure Society of America (HFSA) guidelines committee.
"We are thrilled to have Dr. Walsh take a leadership role on our pivotal study," said Allan Will, chairman and CEO of EBR Systems. "She is an accomplished leader in the Heart Failure therapy community and a widely recognized clinical researcher. By joining this study she is helping illuminate the under-served population of heart failure patients who have been unable to receive or who have not responded to conventional CRT and risk further deterioration or death."
"I am so excited to participate in this very important SOLVE CRT study, and join a group of international experts to evaluate and confirm the promising early results of the WiSE CRT System," said Dr. Walsh. "WISE CRT's endocardial pacing technology has the potential to play a critical role in improving the lives of heart failure patients who have been unable to receive effective care to date and face progressive worsening of their condition."
About Heart Failure and Cardiac Resynchronization Therapy (CRT)
Heart failure is a serious condition in which the heart is unable to pump enough blood to meet the body's demands. A progressive, debilitating disease, heart failure often occurs when electrical signals within the heart are disrupted, causing the heart's ventricles to beat in an uncoordinated or unsynchronized pattern, which in turn enlarges the left ventricle and makes the heart less efficient.
Without therapy, people with heart failure deteriorate and may eventually die of the condition. Cardiac resynchronization therapy offers a proven treatment that improves symptoms and reduces hospitalizations and mortality1,2 by electrically stimulating the heart. Also referred to as biventricular pacing, CRT uses wire leads to synchronize the left and the right ventricles so that the two chambers beat together, thereby improving the heart's efficiency. Approximately 200,000 patients are treated worldwide each year with conventional CRT and about 155,000 of them are in the U.S.3
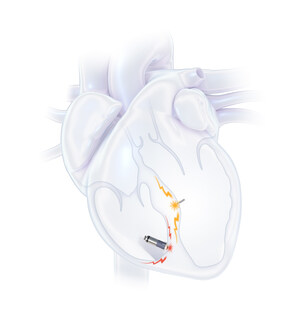
EBR Systems' WiSE Technology
Approximately 30 percent of patients receiving conventional CRT do not respond to the therapy. A major cause of this shortcoming is believed to be the inconsistency of results achieved using wire leads to pace the left ventricle from within the coronary sinus vein on the epicardial (exterior) surface of the heart. Although it is generally accepted that stimulation of the left ventricle is preferable from inside the heart (endocardially), wire leads placed inside the left ventricle can cause clots, heart attacks or strokes. Wired leads can also break, dislodge or otherwise fail, leading to complications in roughly 12 percent of cases.4 Additionally, some patients suffer increased rates of infections and other complications upon the replacement of a cardiac stimulation device.5 The financial impact of these issues is profound. As much as $1 billion of $3.5 billion spent annually on CRT devices provides no patient benefit.4
EBR Systems' WiSE CRT System is the world's only wireless, endocardial (inside the heart) pacing system in clinical use for stimulating the heart's left ventricle. This has long been a goal of cardiac pacing companies since internal stimulation of the left ventricle is thought to be a potentially superior, more physiologic pacing location. WiSE Technology enables cardiac pacing of the left ventricle with a novel cardiac implant that is roughly the size of a large grain of rice. The need for a pacing wire on the outside of the heart's left ventricle – and the attendant problems – are potentially eliminated.
An investigational device in the U.S., the WiSE™ CRT System received European CE Mark approval for its second-generation wireless transmitter and has enabled treatment for many patients who had previously failed treatment with conventional CRT devices.
About EBR Systems
Silicon Valley-based EBR Systems is dedicated to superior treatment of cardiac rhythm disease by providing more physiologically effective stimulation through wireless cardiac pacing. The company's patented proprietary Wireless Stimulation Endocardially (WiSE) technology was developed to eliminate the need for cardiac pacing leads, historically the major source of complications and reliability issues in cardiac rhythm disease management. The company's initial product is designed to eliminate the need for coronary sinus leads to stimulate the left ventricle in heart failure patients requiring CRT. Future products will address wireless endocardial stimulation for bradycardia and other non-cardiac indications. EBR Systems is backed by respected private equity investors including M.H. Carnegie & Co., Brandon Capital Partners, Split Rock Partners, Ascension Ventures, and Emergent Medical Partners.
Contact:
Chris Pfaff
EBR Systems, Inc.
[email protected]
1 McAlister FA, Ezekowitz J, Hooton, N, et al. Cardiac Resynchronization Therapy for Patients with Left Ventricular Systolic Dysfunction: A Systematic Review. JAMA. 2007; 297(22): 2502-14.
2 Abraham WT, Fisher WG, Smith AL, et al. Cardiac Resynchronization in Chronic Heart Failure. N Engl J Med. 2002; 346: 1845-53.
3 JP Morgan, Cardiac Rhythm Management (CRM) Market Model, August, 2017, Michael Weinstein
4 Leon AR, Abraham WT, Curtis AB et.al. Safety of Transvenous Cardiac Resynchronization System Implantation in Patients with Chronic Heart Failure. J Am Coll Cardiol. 2005; 46: 2348-56.
5 Poole JE, Gleva MJ, Mela T, et.al. Complication Rates Associate with Pacemaker or ICD Generator Replacements and Upgrade Procedures. Circulation. 2010;122:1553-1561.
SOURCE EBR Systems







Share this article