Search Results
Mar 19, 2026, 07:30 ET Biohaven's Phase 2 Obesity Study with Taldefgrobep Alfa, a Novel Myostatin-Activin Pathway Inhibitor, Completes Enrollment
realized until four weeks beyond the final dose was administered, suggesting the drug may support extended dosing intervals. In the Phase 3 study BHV2000-301, examining patients with a neuromuscular disorder, taldefgrobep-treated participants achieved statistically significant reductions in total body
More news about: Biohaven Ltd.
Mar 07, 2026, 17:51 ET TTW Picks Best 50 Heritage Travel Destinations in the World for 2026
Karachi Jinnah International Airport (KHI)Baltit Fort, Hunza – Gilgit Airport (GIL)Derawar Fort – Bahawalpur Airport (BHV) / Multan Airport (MUX)Chaukhandi Tombs, Karachi Region – Karachi Jinnah International Airport (KHI)Buddhist Ruins of Takht-i-Bahi –
More news about: Travel And Tour World
Mar 02, 2026, 16:05 ET Biohaven Reports Recent Business Developments and Fourth Quarter and Full Year 2025 Financial Results
applications include BHV-1420 in membranous nephropathy, BHV-1450 in pemphigus vulgaris, myasthenia gravis, and encephalitis, BHV-1440 in Graves' disease and thyroid eye disease, BHV-6500 in type 1 diabetes, BHV-1490 in cryoglobulinemia, Waldenstrom's macroglobulinemia and IgM neuropathy, BHV-1310 for management
More news about: Biohaven Ltd.
Jan 12, 2026, 11:15 ET Biohaven Highlights Portfolio Progress, Positive Early Patient Data from Priority Degrader Programs and Anticipated Milestones at the 44th Annual J.P. Morgan Healthcare Conference
BHV-1955 targeting the oxytocin receptor centrally for the treatment of tinnitus;BHV-2120, oral small molecule TRPM3 inhibitor for epilepsyBHV-8555, a brain penetrant, oral small molecule preventing a-synuclein aggregation in Parkinson's disease; andBHV-8100, a brain-penetrant,
More news about: Biohaven Ltd.
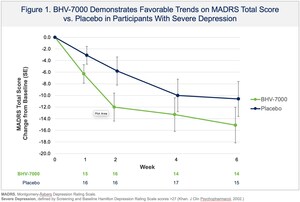
Dec 24, 2025, 13:00 ET Biohaven Provides Update From Phase 2 Proof-of-Concept Study with BHV-7000 in Major Depressive Disorder
expansion cohorts BHV-1400 for IgAN and BHV-1300 for Graves' disease;expectations for the company's recently initiated Phase 2b study with taldefgrobep alfa in obesityoncology clinical stage assets;and emerging data from its ongoing clinical trial with BHV-7000 in adult focal
More news about: Biohaven Ltd.
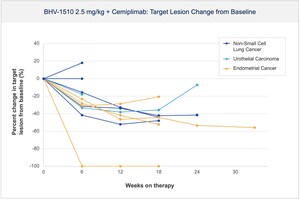
Dec 11, 2025, 07:00 ET Biohaven Presents Clinical Safety and Efficacy Data for BHV-1510, a Next-Generation Trop2 Antibody Drug Conjugate in Combination with Cemiplimab at the 2025 European Society for Medical Oncology (ESMO) Immuno-Oncology Congress
a total of 31 participants were treated with the combination of BHV-1510 and cemiplimab, with BHV-1510 doses ranging from 2-2.75 mg/kg Q3W and 1.25-1.5 mg/kg D1D8Q3W. Cemiplimab was given at 350 mg Q3W.Across all doses BHV-1510, was generally well tolerated with a safety profile differentiated
More news about: Biohaven Ltd.
Nov 10, 2025, 06:58 ET Biohaven Reports Third Quarter 2025 Financial Results and Recent Business Developments
continued progress across our key programs and clinical-stage assets including: MoDE™ and TRAP™ degrader programs, where our two lead assets, BHV-1300 and BHV-1400, show compelling evidence to change the treatment paradigm in immune-mediated diseases; opakalim, a novel Kv7 ion channel activator, for the
More news about: Biohaven Ltd.
Nov 04, 2025, 20:06 ET FDA Issues Complete Response Letter for Biohaven's VYGLXIA (troriluzole) New Drug Application for Spinocerebellar Ataxia
Key areas of focus over the near term include: 1) Clinical-stage, lead extracellular degraders for IgA nephropathy (BHV-1400) and Graves' disease (BHV-1300); 2) Opakalim, Kv7 ion channel activator, pivotal trials in adult focal epilepsy and depression; and 3) Taldefgrobep alfa, myostatin-activin
More news about: Biohaven Ltd.
Sep 24, 2025, 17:31 ET Moderate-to-Severe Acute Pain Market Anticipates Impressive Growth Trajectory at a CAGR of 14% During the Study Period (2020-2034) | DelveInsight
Vocacapsaicin (CA-008), ATX101, HT-6184, TK-254Rx (esflurbiprofen transdermal), CT001, Alenura (VNX001), BHV-2100, and others.By 2034, among all the therapies, the highest revenue is expected to be generated by NURTEC ODT/VYDURA (rimegepant),
More news about: DelveInsight Business Research, LLP
Sep 12, 2025, 08:45 ET Biohaven Ltd. Sued for Securities Law Violations - Investors Should Contact Levi & Korsinsky Before September 12, 2025 to Discuss Your Rights - BHVN
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: Levi & Korsinsky, LLP
Sep 11, 2025, 19:40 ET BHVN Deadline: BHVN Investors Have Opportunity to Lead Biohaven Ltd. Securities Fraud Lawsuit
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (2) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (3) all the foregoing, once revealed, was likely to
More news about: THE ROSEN LAW FIRM, P. A.
Sep 11, 2025, 08:45 ET Shareholders that lost money on Biohaven Ltd.(BHVN) Urged to Join Class Action - Contact The Gross Law Firm to Learn More
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: The Gross Law Firm
Sep 09, 2025, 09:00 ET Levi & Korsinsky Reminds Biohaven Ltd. Investors of the Pending Class Action Lawsuit with a Lead Plaintiff Deadline of September 12, 2025 - BHVN
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: Levi & Korsinsky, LLP
Sep 08, 2025, 14:50 ET BHVN Investors Have Opportunity to Lead Biohaven Ltd. Securities Fraud Lawsuit with the Schall Law Firm
market. Biohaven overstated the regulatory prospects of its drug candidate troriluzole as a treatment for SCA. The Company also overstated the efficacy of BHV-7000 as a treatment for bipolar disorder. Based on these facts, the Company's public statements were false and materially misleading throughout the class
More news about: The Schall Law Firm
Sep 08, 2025, 14:30 ET Pomerantz Law Firm Announces the Filing of a Class Action Against Biohaven Ltd. and Certain Officers - BHVN
is developing, among other product candidates, troriluzole for the treatment of spinocerebellar ataxia ("SCA"), among other indications, as well as BHV-7000 for the treatment of bipolar disorder, among other indications. In May 2022, a Phase 3 trial evaluating troriluzole's
More news about: Pomerantz LLP
Sep 08, 2025, 12:00 ET Biohaven Ltd. (BHVN) Investors Who Lost Money Have Opportunity to Lead Securities Fraud Lawsuit
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (2) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (3) all the foregoing, once revealed, was likely to
More news about: The Law Offices of Frank R. Cruz, Los Angeles
Sep 08, 2025, 08:45 ET The Gross Law Firm Announces the Filing of a Securities Class Action on Behalf of Biohaven Ltd.(BHVN) Shareholders
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: The Gross Law Firm
Sep 06, 2025, 20:40 ET BHVN Deadline: BHVN Investors Have Opportunity to Lead Biohaven Ltd. Securities Fraud Lawsuit
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (2) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (3) all the foregoing, once revealed, was likely to
More news about: THE ROSEN LAW FIRM, P. A.
Sep 06, 2025, 10:03 ET INVESTOR DEADLINE APPROACHING: Faruqi & Faruqi, LLP Investigates Claims on Behalf of Investors of Biohaven
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (2) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (3) all the foregoing, once revealed, was likely to
More news about: Faruqi & Faruqi, LLP
Sep 05, 2025, 08:45 ET Levi & Korsinsky Notifies Biohaven Ltd. Investors of a Class Action Lawsuit and Upcoming Deadline - BHVN
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: Levi & Korsinsky, LLP
Sep 04, 2025, 08:45 ET Contact The Gross Law Firm by September 12, 2025 Deadline to Join Class Action Against Biohaven Ltd.(BHVN)
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: The Gross Law Firm
Sep 03, 2025, 10:00 ET Pomerantz Law Firm Announces the Filing of a Class Action Against Biohaven Ltd. and Certain Officers - BHVN
is developing, among other product candidates, troriluzole for the treatment of spinocerebellar ataxia ("SCA"), among other indications, as well as BHV-7000 for the treatment of bipolar disorder, among other indications. In May 2022, a Phase 3 trial evaluating troriluzole's
More news about: Pomerantz LLP
Sep 02, 2025, 12:00 ET Biohaven Ltd. (BHVN) Investors Who Lost Money Have Opportunity to Lead Securities Fraud Lawsuit
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (2) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (3) all the foregoing, once revealed, was likely to
More news about: Glancy Prongay & Murray LLP
Sep 02, 2025, 08:45 ET Levi & Korsinsky Notifies Biohaven Ltd. Investors of a Class Action Lawsuit and Upcoming Deadline - BHVN
and/or the sufficiency of data that Biohaven submitted in support of troriluzole's regulatory approval for this indication, were overstated; (ii) BHV-7000's efficacy and clinical prospects as a treatment for bipolar disorder were likewise overstated; (iii) all the foregoing, once revealed, was likely
More news about: Levi & Korsinsky, LLP
Sep 02, 2025, 03:30 ET BHVN Investors Have Opportunity to Lead Biohaven Ltd. Securities Fraud Lawsuit with the Schall Law Firm
market. Biohaven overstated the regulatory prospects of its drug candidate troriluzole as a treatment for SCA. The Company also overstated the efficacy of BHV-7000 as a treatment for bipolar disorder. Based on these facts, the Company's public statements were false and materially misleading throughout the class
More news about: The Schall Law Firm