
BD Announces Second FDA Emergency Use Authorization, CE Mark for New COVID-19 Molecular Diagnostic for Global Use
New Test Uses BD MAX™ System to Augment Supply from Existing Collaborations; Test Results in Under Three Hours
FRANKLIN LAKES, N.J., April 13, 2020 /PRNewswire/ -- BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced that the U.S. Food and Drug Administration (FDA) has granted Emergency Use Authorization (EUA) for an additional molecular diagnostic test for COVID-19 that can return results in two to three hours. The new test also has been CE marked to the IVD Directive (98/79/EC).
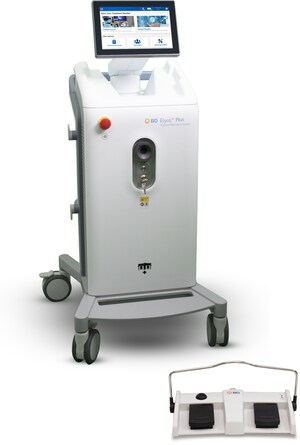
The test, which is run on the BD MAX™ System, provides additional testing capacity for COVID-19 in the United States and in countries that recognize the CE Mark to test patients and health care workers. The test is in addition to the other tests already available on the BD MAX™ System from collaborations with BioGX and CerTest and is based on the CDC assay design. The BD MAX™ System, a molecular diagnostic platform, is already in use at thousands of laboratories worldwide, and each unit is capable of analyzing hundreds of samples over a 24-hour period.
"The new BD COVID-19 test for the BD MAX™ System will help increase availability of these much-needed tests around the world," said Dave Hickey, president of Integrated Diagnostics for BD. "We continue to work towards a full portfolio of testing options to give health care workers choice and access to the right test for the right situation."
The majority of BD MAX™ Systems are installed in hospital laboratories, reducing the added time and complexity of needing to send samples to a reference lab. The system is fully automated, reducing the opportunity for human error and increasing the speed to result, and can process 24 samples simultaneously. The assay is based on the same viral RNA targeting sequences and real-time PCR detection method as the test developed by the U.S. Centers for Disease Control and Prevention (CDC).
Hospitals and laboratories that use a BD MAX™ System can order tests through their BD sales representative.
The BD SARS-CoV-2 Reagent Kit for BD MAX™ System has been CE marked to the IVD Directive (98/79/EC), but it has not been cleared or approved by FDA. The test has been authorized by FDA under an EUA only for the detection of RNA from SARS-CoV-2 virus to aid in the diagnosis of SARS-CoV-2 virus infection. It has not been authorized for use to detect any other viruses or pathogens. The test is authorized in the United States for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostic tests for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
About BD
BD is one of the largest global medical technology companies in the world and is advancing the world of health by improving medical discovery, diagnostics and the delivery of care. The company supports the heroes on the frontlines of health care by developing innovative technology, services and solutions that help advance both clinical therapy for patients and clinical process for health care providers. BD and its 65,000 employees have a passion and commitment to help enhance the safety and efficiency of clinicians' care delivery process, enable laboratory scientists to accurately detect disease and advance researchers' capabilities to develop the next generation of diagnostics and therapeutics. BD has a presence in virtually every country and partners with organizations around the world to address some of the most challenging global health issues. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase efficiencies, improve safety and expand access to health care. For more information on BD, please visit bd.com.
Contacts: |
|
Troy Kirkpatrick |
Monique N. Dolecki |
BD Public Relations |
BD Investor Relations |
858.617.2361 |
201.847.5378 |
SOURCE BD (Becton, Dickinson and Company)







Share this article