News Search Results
Mar 31, 2026, 16:45 ET Kodiak Sciences Announces Recent Business Highlights and Fourth Quarter and Full Year 2025 Financial Results
over time in wet AMD patients treated with anti-VEGF monotherapy, which suggests that IL-6 inhibition in combination with anti-VEGF therapy could lead to improved outcomes.Kodiak has advanced KSI-501 into the Phase 3 study DAYBREAK to evaluate its efficacy and safety in wet AMD. DAYBREAK has completed
More news about: Kodiak Sciences Inc.
Mar 26, 2026, 06:00 ET Kodiak Sciences Announces Positive Topline Results in GLOW2, the Second Phase 3 Study in Diabetic Retinopathy, Demonstrating Superiority of Zenkuda™ (tarcocimab tedromer) Over Sham
over time in wet AMD patients treated with anti-VEGF monotherapy, which suggests that IL-6 inhibition in combination with anti-VEGF therapy could lead to improved outcomes.Kodiak has advanced KSI-501 into the Phase 3 study DAYBREAK to evaluate its efficacy and safety in wet AMD. DAYBREAK has completed
More news about: Kodiak Sciences Inc.
Mar 11, 2026, 11:45 ET REGENXBIO REPORTS NEW POSITIVE INTERIM DATA FROM PHASE I/II AFFINITY DUCHENNE® TRIAL OF RGX-202
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Mar 05, 2026, 07:05 ET REGENXBIO Reports Fourth Quarter and Full Year 2025 Financial Results and Operational Highlights
collaboration with AbbVie, is potentially the first-in-class treatment for wet age-related macular degeneration (wet AMD) and diabetic retinopathy (DR).Sura-vec for the Treatment of Wet AMD (Subretinal Delivery)REGENXBIO expects to share topline data with AbbVie from ATMOSPHERE®
More news about: REGENXBIO Inc.
Mar 04, 2026, 07:05 ET REGENXBIO Announces Presentations at the 2026 Muscular Dystrophy Association (MDA) Clinical & Scientific Conference
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Mar 02, 2026, 07:05 ET REGENXBIO to Participate in Upcoming Investor Conferences
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Feb 25, 2026, 10:45 ET Age Related Macular Degeneration Market to Reach $16.2 billion by 2033, Globally, by 2034 at 5% CAGR: Allied Market Research
Age related macular degeneration Market by Product Type (Aflibercept, Ranibizumab and Others), Disease Type (Dry AMD and Wet AMD) and Distribution Channel (Hospital Pharmacy, Specialty Pharmacy and Online Pharmacy): Global Opportunity Analysis and Industry Forecast, 2022-2032".
More news about: Allied Market Research
Feb 25, 2026, 07:05 ET REGENXBIO to Host Conference Call on March 5 to Discuss Fourth Quarter and Full Year 2025 Financial Results and Operational Highlights
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Feb 09, 2026, 16:05 ET REGENXBIO Announces Regulatory Update on RGX-121 BLA for MPS II
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Jan 29, 2026, 04:00 ET LifeArc Ventures 2025: New geographies and investments, a growing team and continued portfolio progress
IKAR-001, first-in-class dual pathway gene therapy for geographic atrophy, and promising data presented on therapeutic potential in geographic atrophy and wet AMD.Maxion Therapeutics' leadership and Board strengthened: Joel Edwards (formerly VP of Corporate Strategy and Operations
More news about: LifeArc
Jan 28, 2026, 07:30 ET REGENXBIO Announces Regulatory Update on Ultra Rare MPS Programs
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Jan 27, 2026, 09:30 ET February is Age-related Macular Degeneration (AMD) and Low Vision Month at Prevent Blindness, Designed to Provide Helpful Sight-saving Resources and Support Programs
geographic atrophy.Wet- Wet AMD generally causes more rapid and more serious vision loss. In this form of the disease, tiny new blood vessels grow under and into the retina. These
More news about: Prevent Blindness
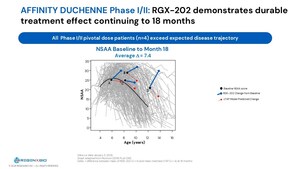
Jan 11, 2026, 15:00 ET REGENXBIO Highlights Key 2026 Catalysts and Announces Positive Long-Term Functional Outcomes in Lead Duchenne Gene Therapy Program
macular degeneration (wet AMD) and diabetic retinopathy (DR)Sura-vec is being developed in collaboration with AbbVie, and could be the first gene therapy for a non-rare disease, if approved.Sura-vec is on track to be the first gene therapy for wet AMD. REGENXBIO expects
More news about: REGENXBIO Inc.
Jan 08, 2026, 19:00 ET Innovent's Partner Ollin Biosciences Announces Positive Topline Data with Superior Outcomes from a Randomized Head-to-Head Study of IBI324 Compared to Faricimab (Vabysmo®) in Diabetic Macular Edema and Wet Age-Related Macular Degeneration
Ehrlich added, "We look forward to discussing these data with health authorities and moving OLN324 quickly into global Phase 3 studies in both DME and wet AMD."In a previously-completed Phase 1 single and multiple dose escalation study, IBI324 (OLN324) demonstrated promising vision and anatomic improvements
More news about: Innovent Biologics
Dec 18, 2025, 07:05 ET REGENXBIO to Present at the 44th Annual J.P. Morgan Healthcare Conference
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Nov 25, 2025, 07:05 ET REGENXBIO to Participate in Upcoming Investor Conference
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Nov 24, 2025, 12:00 ET Merck Manuals Answers Common Questions About Age-Related Macular Degeneration
result, central vision becomes washed out and loses detail, and straight lines may appear wavy.What is the difference between dry and wet AMD?There are 2 types of AMD: dry and wet. All AMD starts as the dry type, says Mehta. In dry AMD, which is characterized by the drusen deposits,
More news about: The Merck Manuals
Nov 13, 2025, 16:05 ET Kodiak Sciences Announces Recent Business Highlights and Third Quarter 2025 Financial Results
Phase 3 DAYBREAK wet AMD study – topline data expected 3Q 2026 KSI-501 Phase 3 DAYBREAK wet AMD study – topline data expected
More news about: Kodiak Sciences Inc.
Nov 07, 2025, 08:00 ET Shinobi Therapeutics Announces the Appointment of Dr. Laurent Fischer as Chair of its Board of Directors
most recently, the sale of Adverum Biotechnologies to Eli Lilly in October 2025, handing off an advanced and potentially curative gene therapy for wet AMD. Dr. Fischer brings extensive board experience, including current positions as first independent Director of Lycia Therapeutics,
More news about: Shinobi Therapeutics
Nov 06, 2025, 07:05 ET REGENXBIO Reports Third Quarter 2025 Financial Results and Operational Highlights
AbbVie, is potentially the first-in-class treatment for wet age-related macular degeneration (wet AMD) and diabetic retinopathy (DR). Sura-vec for the Treatment of Wet AMD (Subretinal Delivery)
More news about: REGENXBIO Inc.
Oct 30, 2025, 07:05 ET REGENXBIO Announces Completion of Pivotal Enrollment and Initiates Commercial Production in Duchenne Gene Therapy Program
MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV
More news about: REGENXBIO Inc.
Oct 30, 2025, 04:16 ET 新橋生物子公司 Visara 將 VIS-101 在大中華區及部分亞洲國家的獨家許可轉讓給雲頂新耀
是一款靶向 VEGF-A/ANG2 的新型雙特異性生物分子,相較現行標準療法,有望為 wet AMD 、 DME 及 RVO
More news about: 新橋生物
Oct 29, 2025, 07:05 ET REGENXBIO to Host Conference Call on November 6 to Discuss Third Quarter 2025 Financial Results and Operational Highlights
II and RGX-111 for the treatment of MPS I, both in partnership with Nippon Shinyaku; and surabgene lomparvovec (ABBV-RGX-314) for the treatment of wet AMD and diabetic retinopathy, in collaboration with AbbVie. Thousands of patients have been treated with REGENXBIO's AAV platform, including those receiving
More news about: REGENXBIO Inc.
Oct 24, 2025, 08:30 ET Lilly to Acquire Adverum Biotechnologies
compliance, and improve vision outcomes for patients with wet AMD. In recognition of the need for new treatment options for wet AMD, FDA granted Fast Track and Regenerative Medicine Advanced Therapy (RMAT) designations for Ixo-vec for the treatment of wet AMD. Ixo-vec has also received PRIME designation from
More news about: Eli Lilly and Company
Oct 23, 2025, 08:00 ET Astellas and the Internet's Favorite Grandma, Barbara 'Babs' Costello, Partner to Encourage Others to Get Ahead of Geographic Atrophy (GA)
small specks floating in your vision, flashes of light, or increased sensitivity to light. There is a risk of developing wet AMD with IZERVAY. You should report any symptoms (visual distortions such as straight lines seeming bent, deterioration in vision, dark spots, loss of
More news about: Astellas Pharma Inc.