Search Results
Mar 16, 2026, 02:00 ET Roche receives CE mark for first IVD immunoassay blood test to identify carriers of ApoE4, a genetic variant present in up to 60% of patients diagnosed with Alzheimer's disease
https://www.who.int/news/item/07-12-2017-dementia-number-of-people-affected-to-triple-in-next-30-years. - Accessed in April 2025[4] Jack CR Jr, Andrews JS, Beach TG, et al. Revised criteria for diagnosis and staging of Alzheimer's disease: Alzheimer's Association Workgroup. Alzheimers Dement
More news about: Roche
Mar 16, 2026, 02:00 ET Roche receives CE mark for first IVD immunoassay blood test to identify carriers of ApoE4, a genetic variant present in up to 60% of patients diagnosed with Alzheimer's disease
https://www.who.int/news/item/07-12-2017-dementia-number-of-people-affected-to-triple-in-next-30-years. - Accessed in April 2025[4] Jack CR Jr, Andrews JS, Beach TG, et al. Revised criteria for diagnosis and staging of Alzheimer's disease: Alzheimer's Association Workgroup. Alzheimers Dement
More news about: Roche
Mar 16, 2026, 00:54 ET Exicom Inaugurates Integrated Manufacturing Facility in Hyderabad to Scale EV Charging and Critical Power Solutions
Built with an investment of INR 216 cr, the facility reflects Exicom's long-term commitment to localized manufacturing and building globally competitive technology infrastructure from India.
More news about: Exicom Tele-Systems Ltd.
Mar 13, 2026, 12:35 ET Johnson & Johnson highlights promising first-in-human Erda-iDRS (formerly TAR-210) results in intermediate-risk non-muscle-invasive bladder cancer
bladder cancer had received treatment. The primary endpoint was safety, with the secondary endpoints assessing complete response rate and duration of CR in the intermediate-risk cohort and recurrence-free survival in the high-risk cohort.1In the intermediate-risk cohort, Erda-iDRS was
More news about: Johnson & Johnson
Mar 13, 2026, 11:00 ET Say Goodbye to Clutter and Hassle: VEVOR Four-Piece Set Transforms Your Garage
VEVOR's impact socket set is built to handle high-torque jobs. Premium CR-V alloy steel and a six-point design grip bolts securely, so even the toughest fasteners come loose without slipping. Multiple socket sizes and extension
More news about: Vevor
Mar 12, 2026, 14:21 ET Steele Honda Announces Special Incentives During Drive Home A Winner Days Event
reduce fuel consumption.Drivers seeking a versatile and efficient SUV can also explore the 2026 Honda CR-V Hybrid, which is currently available with lease rates starting at 2.99%. The CR-V Hybrid offers spacious interior comfort, advanced safety technology, and the capability families and commuters
More news about: Steele Honda
Mar 09, 2026, 11:43 ET Tarrytown Honda Earns Honda President's Award
with the HR-V, CR-V, Passport, Prologue and Pilot sport utility vehicles, the Ridgeline pickup and the Odyssey minivan. The Honda electrified vehicle lineup, representing more than a quarter of total sales in 2025, includes the all-electric Prologue SUV, hybrid-electric Accord, CR-V, and Civic, and Fuel-Cell-electric
More news about: Tarrytown Honda
Mar 06, 2026, 08:55 ET HONEYWELL ANNOUNCES COMMENCEMENT OF CASH TENDER OFFERS TO PURCHASE UP TO $3,750,000,000 AGGREGATE PURCHASE PRICE OF DOLLAR-DENOMINATED SECURITIES AND UP TO €1,250,000,000 AGGREGATE PURCHASE PRICE OF EURO-DENOMINATED SECURITIES
2031CUSIP: 438516CR5ISIN: US438516CR55September 1, 2031
More news about: Honeywell
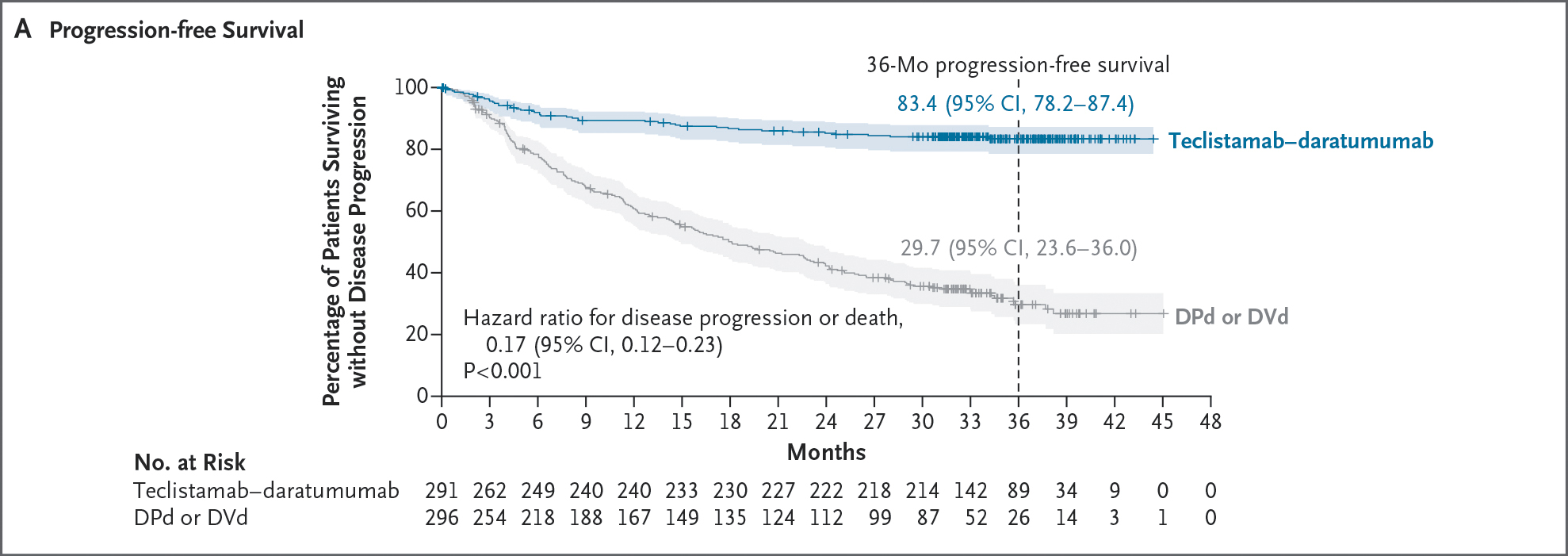
Mar 05, 2026, 15:55 ET Johnson & Johnson Announces U.S. FDA Approval of TECVAYLI® plus DARZALEX FASPRO® for Relapsed/Refractory Multiple Myeloma, Offering a Potential New Standard of Care as Early as Second Line
DARZALEX FASPRO® showed higher rates of overall response (ORR) (89.0% vs. 75.3%; OR, 2.65; 95% CI, 1.68-4.18), complete response (≥CR) (81.8% vs. 32.1%; odds ratio [OR], 9.56; 95% CI, 6.47-14.14), and MRD-negativity (58.4% vs. 17.1%; OR, 6.78; 95% CI, 4.53-10.15, P<0.0001; evaluable
More news about: Johnson & Johnson
Mar 05, 2026, 13:30 ET Newly Listed Fractal Crosses Rs 100 Cr PAT in Q3
expansion of 0.2% YoY. Adjusted EBITDA outperformed revenue growth, clocking 24% growth YoY to Rs 152.1 Cr. Adjusted EBITDA margin was at 17.8%, an expansion of 0.4% YoY. Profit after Tax was Rs 100.1 Cr. Fractal Products beating Global BenchmarksThe company's consumer-targeted
More news about: Fractal Analytics Limited
Mar 05, 2026, 11:08 ET Minnesota Medical Technologies Corporation Secures $20 Million in Series A Funding to Accelerate Growth
experienced team with a proven track record of value creation, including leadership experience from Rochester Medical Corporation (sold in 2013 for $262M to CR Bard), Minnesota Medical Technologies Corporation is committed to developing, making, and selling Continence Care Products of unmatched comfort, performance,
More news about: Minnesota Medical Technologies
Mar 05, 2026, 09:00 ET YSL BEAUTY UNVEILS NEW CAST OF US LOCAL AMBASSADORS: LILA MOSS, AMELIA GRAY, AND LAURA HARRIER
going through adolescence. Her commercial fashion photography has been published in dozens of publications such as Vogue, Elle, Harper's Bazaar, GQ, CR Men, and W. She has shot campaigns for various luxury brands as well as photographing the cover image of Kendrick Lamar's album, Mr. Morale & The
More news about: YSL Beauty
Mar 04, 2026, 21:26 ET Cadonilimab Achieves 100% 24-Month OS in Complete Responders in R/M Cervical Cancer Based on Long-Term Phase II Results
was 47.3% (nominal p < 0.0001).The median time to response (mTTR) in the CR patients was 1.84 months, comparable to that observed in the PR patients (1.87 months). The median duration of response (mDoR) in the CR patients was not reached and was significantly longer than that in the PR patients
More news about: Akeso, Inc.
Mar 04, 2026, 13:20 ET /C O R R E C T I O N -- Create Music Group/
independent electronic music labels with 1000's of releases and a global fan base; !K7 Music, a Berlin-based label group with deep cultural heritage; Cr2 Records, a UK dance music platform encompassing label, publishing and creative tools businesses; and Mau5trap the iconic label founded by
More news about: Create Music Group
Mar 04, 2026, 12:06 ET Huawei, Otonom Ağ İşletme ve Bakımı Alanında Bir Çığır Açarak Sektörün İlk Kampüs L4 ADN Çözümünü Tanıttı
content:https://www.prnewswire.com/news-releases/huawei-otonom-a-iletme-ve-bakm-alannda-bir-cr-acarak-sektorun-ilk-kampus-l4-adn-cozumunu-tantt-302704212.html
More news about: Huawei
Mar 04, 2026, 09:15 ET Citius Oncology Announces Preliminary Topline Phase 1 Data from Study of LYMPHIR™ (E7777) Dosing Prior to Commercial CAR‑T Therapy in High‑Risk Diffuse Large B‑Cell Lymphoma
investigator‑initiated study at the University of Minnesota and City of Hope demonstrates 86% overall response rate (OR), including 57% complete response (CR) and 29% partial response (PR)LYMPHIR was well-tolerated with no dose-limiting toxicities observedCRANFORD,
More news about: Citius Oncology, Inc.
Mar 03, 2026, 08:00 ET Stryker showcases continued innovation across orthopaedics at AAOS 2026 Annual Meeting
doi:10.2106/jbjs.18.01162Stryker Test Report: D0000325745 Triathlon Gold Micromotion Verification Testing. July, 2025.Stryker Test Report: D0000321183. Triathlon Gold CR Femoral Component: Posterior Femoral Fatigue Verification Testing. September, 2025.Stryker Test Report: D0000394012. Comparative Evaluation
More news about: Stryker
Mar 02, 2026, 08:30 ET XTI Aerospace's Drone Nerds Business Expands Creality Line with New SPARKX i7 3D Printer
worldwide. Today, the flagship K series continues to push the limits of next-generation maker technology. Additional product lines include the Ender, Hi, and CR series. Creality's in-house developed software, such as Creality Print and Creality Cloud App, creates a seamless workflow across devices. With a comprehensive
More news about: XTI Aerospace, Inc.
Feb 27, 2026, 11:14 ET ¡Más deporte, más adrenalina! Samsung TV Plus enciende la emoción con transmisiones en vivo y gratis
2022. A unos meses de celebrar un nuevo Mundial en México, podrás revivir todos los goles históricos de la Selección Mexicana y leyendas como Ronaldo, CR7 y Messi.El canal también transmite películas y series originales con emotivas historias de vida que nos dejan ver el lado más humano de cracks
More news about: Samsung Mexico
Feb 27, 2026, 08:34 ET Canada Nickel Announces Filing of NI 43-101 Technical Report for Previously Announced Updated Resource, Reid Nickel Sulphide Project
an effective date of January 7, 2026, are titled "National Instrument 43-101 Technical Report and Updated Mineral Resource Estimate for the Reid Ni-Co-Cr-PGE Deposit, Reid Nickel Sulphide Project." The Reports was prepared for Canada Nickel by Caracle Creek International Consulting Inc. and can be found
More news about: Canada Nickel Company Inc.
Feb 26, 2026, 16:00 ET Federal Signal Corporation Appoints Two New Directors
experience will make him a valuable addition to our board." Since January 2013, Mr. Maue has served as the CFO of Crane Company ("Crane") (NYSE: CR), a leading manufacturer of highly engineered components for challenging, mission-critical applications focused on the aerospace, defense, space and process
More news about: Federal Signal Corporation
Feb 26, 2026, 09:15 ET E&I Cooperative Services Awards Unimarket Cooperative Contract for Source-to-Pay Solutions, Expanding Access for Education Institutions
global leader in source-to-pay and spend management solutions, announced it has been awarded a competitively solicited contract (Contract Number: CR001502) with
More news about: Unimarket
Feb 25, 2026, 16:15 ET Becton, Dickinson and Company Announces Pricing of the Tender Offers and Amounts Accepted for Purchase
Notes due 2029075887CR8$625,000,000
More news about: BD (Becton, Dickinson and Company)
Feb 25, 2026, 08:00 ET Becton, Dickinson and Company Announces Early Tender Results and Upsizing of Offer SubCap and Aggregate Offer Cap
Notes due 2029075887CR8$625,000,000
More news about: BD (Becton, Dickinson and Company)
Feb 24, 2026, 23:47 ET SuperProcure, a Unified TMS Platform, Secures ₹14 Cr Investment Led by Pentathlon Ventures
multimedia:https://www.prnewswire.com/in/news-releases/superprocure-a-unified-tms-platform-secures-14-cr-investment-led-by-pentathlon-ventures-302696123.html
More news about: SuperProcure